A reagent can look fine today and turn weak next week. Light is often the hidden trigger, and the damage shows up only after results drift.
Chemicals go into amber glass because amber cuts UV and blue light that drives photodegradation, while glass stays inert and seals well. It is the simplest “passive protection” for light-sensitive reagents.

Light is not just “brightness.” Light is energy, and some wavelengths push reactions forward. Many labs learn this the hard way. A bottle of silver nitrate can darken. Some peroxide solutions lose strength. A dye standard can fade. When that happens, the bottle did not “go bad” by accident. It got a slow dose of photons.
Light is a reagent in slow motion
The short wavelengths do the most damage. Ultraviolet radiation 1 starts radical pathways fast. Blue and violet can also excite many organic molecules. Amber glass works because its colorants absorb a large part of that UV-to-blue band before the light reaches the liquid. This matters for reagents that oxidize, polymerize, or isomerize under light. It also matters for solutions with indicators, vitamins, and plant extracts that shift color.
Amber is protection, not a promise
Amber reduces exposure, but it does not make a reagent immortal. Clear storage rules still matter. The cap must seal. The headspace should not pull in humid air. The shelf should stay cool. For very sensitive materials, amber is often paired with a carton, foil wrap, or a closed cabinet.
When amber also helps daily lab work
Amber bottles create a visual “flag.” Everyone can spot light-sensitive chemicals quickly. Labels also last longer because less UV hits the ink. This is a small detail, but it cuts mistakes.
| What can go wrong without amber | What causes it | What amber changes | Extra step when risk is high |
|---|---|---|---|
| Loss of potency | UV/blue driven reactions | Less short-wavelength light reaches the liquid | Store in a closed cabinet or carton |
| Discoloration | Photo-oxidation and radicals | Slows color change | Add secondary opaque wrap |
| Drift in standards | Isomerization or breakdown | Improves stability during routine handling | Reduce headspace, use fresh aliquots |
| Label fading | UV exposure | Extends label life | Use chemical-resistant label stock |
Amber glass is chosen because it is a low-effort control. It reduces a common failure mode without changing the chemistry or the workflow.
The next questions decide how strong that control needs to be, and which closure and glass grade actually match the chemical.
Does amber block UV/blue light that degrades reagents?
A bottle can be “brown” and still let the wrong light through. That is why some reagents fail even in amber glass when they sit under strong lab lighting.
Amber glass is used because it blocks most UV and a big portion of violet/blue light, which are the wavelengths most likely to start photodegradation. It does not block all light, so highly photosensitive reagents may need secondary opaque packaging.
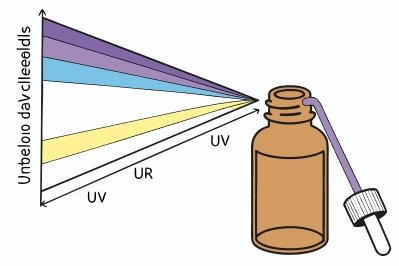
Amber works by absorption, not by reflection. The colorants in the glass absorb short wavelengths, and the glass thickness increases that effect. In practice, amber bottles are often sold as “light-resistant containers,” which is not a casual marketing phrase in regulated industries. Pharmacopeial and guideline-driven stability work often frames this under photostability testing guidance such as ICH Q1B 2 because transmission in the UV/blue region is where many actives absorb energy.
The practical detail is this: “amber” is not one exact formula. Different suppliers can hit different transmission curves, and bottle thickness varies by size. So a small amber vial and a large amber reagent bottle do not always give identical protection. That is also why labs still rely on common-sense habits, like keeping bottles out of direct sunlight and closing cabinets.
A simple way to judge whether amber is enough
If the SDS or monograph says “protect from light,” amber is a baseline. If it says “protect from light and store in outer carton,” amber alone is usually not enough. If the reagent darkens during normal room handling, amber plus secondary protection is needed.
Common “extra protection” options
- outer cartons for routine storage
- foil wrap for bench use
- low-light handling for transfers
- small aliquots to reduce repeated light exposure
| Sensitivity level (practical) | Typical clue | Amber bottle alone | Best add-on control |
|---|---|---|---|
| Mild | slow color change over months | Often enough | Closed cabinet storage |
| Medium | change over weeks under lab lights | Sometimes enough | Carton or amber + cabinet |
| High | change over days or after transfers | Often not enough | Foil wrap + aliquots |
| Extreme | reacts during brief exposure | Not enough | Opaque container + low-light workflow |
Amber blocks a lot of the harmful band, but it still lets longer wavelengths pass. The goal is not darkness. The goal is a proven reduction in the wavelengths that trigger the failure.
Are ground-glass stoppers and PTFE liners required?
A perfect bottle can still fail because the closure is wrong. Leaks, stuck stoppers, and liner swelling cause more trouble than most people expect.
Ground-glass stoppers and PTFE liners are not always required, but closure choice must match the chemical. Ground-glass avoids many polymer interactions, while PTFE-faced liners give broad resistance for solvents and many acids. Some oxidizers and off-gassing chemicals need venting designs.

Ground-glass stoppers are the classic lab solution. They can form a tight seal without a separate gasket, and they avoid direct contact between the liquid and many plastics. That matters for high-purity solvents, trace analysis, and reagents that extract additives from polymers. But ground-glass has two real drawbacks in daily work: it can seize, and it can leak if the joint gets dirty or chipped. Some chemicals also “cement” joints over time, especially when crystals form near the neck—an issue tied to how ground-glass joints 3 seal by surface contact.
Screw caps solve handling problems, but the liner becomes the real contact surface. PTFE-faced liners 4 are popular because PTFE resists a very wide range of chemicals at room temperature. Many labs pair amber glass with phenolic caps and PTFE-faced liners for solvents, strong acids, and general reagents. Still, “broad compatibility” is not the same as “universal.” Rubber liners can be poor with acids or alkalis. Foil/pulp liners can be good with many organics but can fail with corrosives. For oxidizers like hydrogen peroxide, the closure must also manage pressure buildup, so special cap designs or venting features may be used.
A practical closure selection mindset
Choose the closure for the failure mode:
- purity risk → reduce polymer contact
- volatility risk → prioritize vapor-tight sealing
- pressure risk → allow controlled venting, not a loose cap
| Chemical situation | Main risk | Ground-glass stopper | Screw cap + liner choice |
|---|---|---|---|
| High-purity solvents (GC/HPLC) | extractables, contamination | Good, but can seize | PTFE-faced liner is common |
| Strong mineral acids | corrosion, fumes | OK if clean, but handling is slow | PTFE-faced liner on phenolic cap |
| Strong bases | glass attack at extremes, crusting | Sticking is common | PTFE-faced liner, short storage time |
| Oxidizers that off-gas | pressure buildup | Not ideal | Vented cap or supplier-specific closure |
| Routine buffers | evaporation, mix-ups | Overkill | PE/PP caps may be fine, follow SDS |
The closure is part of compatibility. Amber glass protects from light, but the stopper and liner protect from leaks, contamination, and pressure surprises.
How do bottle grades (Type I/II/III) affect compatibility?
Two amber bottles can look identical and still behave differently with water, acids, or heat. The “Type” rating explains why.
Type I, II, and III glass grades describe chemical durability, mainly hydrolytic resistance. Type I (often borosilicate) is the most resistant and is the safest default. Type II is surface-treated soda-lime with improved resistance. Type III is soda-lime and is usually best for dry solids or less demanding storage.

The Type system is used in pharmaceutical and laboratory supply chains because water is a strong test chemical. If glass releases alkali ions into water under test conditions, it signals weaker chemical durability. That matters for injectables and for sensitive standards. Type I is the top tier and is often associated with borosilicate glass 5 compositions, which have high hydrolytic resistance and good thermal performance. Type II begins as soda-lime glass, then receives an internal surface treatment that improves hydrolytic resistance. Type III is untreated soda-lime glass with moderate hydrolytic resistance.
For many routine lab reagents, Type III works fine. That is why it is common in general reagent bottles. But for aggressive aqueous solutions, for long storage, for high-purity analytical work, or for repeated heat cycles, Type I is the safer choice. Compatibility also has hard exceptions. Hydrofluoric acid should not be stored in glass at all 6. Very strong alkaline solutions can also attack glass, especially at elevated temperature, and that can lead to etching and contamination.
A simple decision rule for labs and brands
- If the chemical is valuable, regulated, or analytically sensitive → lean Type I.
- If it is aqueous and the pH is not extreme → Type II can be acceptable.
- If it is a dry solid or low-risk reagent → Type III is often sufficient.
| Glass grade | Common description | Durability level | Best-fit examples | Watch-outs |
|---|---|---|---|---|
| Type I | often borosilicate | Highest | injectables, standards, acids/neutral solutions | avoid HF; strong hot alkali still risky |
| Type II | treated soda-lime | High (surface) | many aqueous drugs, some lab reagents | treatment is on the surface, not bulk |
| Type III | soda-lime | Moderate | dry salts, routine reagents, short storage | not ideal for demanding aqueous purity |
The Type label is not about color. Amber can exist across Types. The Type label is about how the glass behaves when chemistry and time start to push back.
Which labeling and venting rules apply to solvents/acids?
A bottle can be perfectly chosen and still be unsafe if it is mislabeled or stored wrong. Most incidents start with confusion, not chemistry.
For solvents and acids, labels must communicate identity and hazards, and storage must follow flammable and corrosive rules from workplace standards and local codes. Venting is not “leave it loose.” It is controlled pressure relief when a chemical off-gasses, using approved caps and proper storage ventilation.

Labeling has two jobs: tell people what the chemical is, and warn them what it can do. In many workplaces, hazard communication rules require shipped containers to carry standardized label elements. In labs, there is also an important practical split: the original supplier bottle is one thing, and a secondary bottle or beaker used for transfers is another. Many labs still label every secondary container because “unlabeled” becomes “unknown” fast. Best practice is simple and fast: full chemical name, concentration (if applicable), key hazards, date received or opened, and the owner or group—aligned with the OSHA Hazard Communication Standard 7. For very small containers, a short label can work if it clearly links to a full record.
Venting is about pressure and vapor control. Flammable solvents should be stored closed, in approved containers, and in proper flammable storage. Corrosive acids should be stored in corrosives storage with secondary containment and segregation by compatibility. Some chemicals generate gas or build pressure in normal storage. In those cases, a vented cap or a supplier-designed pressure-management closure is safer than a tight seal that can over-pressurize. Still, venting must not create a vapor hazard. The SDS and local EHS rules should decide when a vented closure is allowed and where the bottle must sit.
| Situation | Label must include | Storage rule focus | Venting approach |
|---|---|---|---|
| Flammable solvents | identity + flammability hazard | flammable cabinet limits and ignition control | keep tightly closed; vent cabinet only if required by authority |
| Strong acids | identity + corrosion hazard | corrosives cabinet + trays + segregation | keep closed; vent storage area if required; never mix incompatibles |
| Strong oxidizing acids | identity + oxidizer/corrosive | segregate from organics and reducers | only use approved closures; manage fumes with proper storage ventilation |
| Off-gassing oxidizers (example: peroxides) | identity + oxidizer warning + date opened | cool storage, away from contaminants | use approved pressure-management / vented caps per supplier guidance |
| Secondary containers | identity + quick hazard cue | keep with responsible user or per lab plan | not about venting; it is about clarity and control |
Amber glass supports stability, but safety still depends on labels people can read and storage rules people actually follow. When labeling and venting are handled with the same care as material selection, shelf life improves and incidents drop.
Conclusion
Amber glass protects light-sensitive chemicals by reducing UV/blue exposure, while Type ratings and closure choices handle chemistry, sealing, and pressure. Good labels and controlled venting complete the system.
Footnotes
-
UV spectrum basics and why short wavelengths deliver higher energy to molecules. ↩︎ ↩
-
Read the ICH framework for photostability testing and how light exposure is controlled and evaluated. ↩︎ ↩
-
Quick reference on how ground-glass joints seal—and why contamination or chips cause sticking and leaks. ↩︎ ↩
-
Use a PTFE resistance reference when selecting liners for solvents, acids, and oxidizers. ↩︎ ↩
-
Background on borosilicate chemistry and why it improves durability in demanding lab storage. ↩︎ ↩
-
Safety reference for hydrofluoric acid, including properties and handling considerations relevant to container choice. ↩︎ ↩
-
Official HazCom overview explaining required label elements, pictograms, and workplace hazard communication expectations. ↩︎ ↩








