Seeds and color drift can wreck a whole run. Scrap rises, the line slows, and every department points at the furnace.
Trace Sb₂O₃ is added because it helps remove bubbles (fining) and helps keep melt redox steady, which supports consistent color—especially in clear glass—when raw materials, cullet, and furnace conditions shift.

The link between Sb₂O₃, redox, and “quiet” glass
Antimony oxide sits in a special category of glass additives. It is not there to build the network like SiO₂. It is not there to lower melt temperature like Na₂O. It is there because it can change oxidation state in the melt. That single feature touches two painful problems at once: bubbles and color.
What “trace” really means in production
In most container recipes, fining and color-control additives are tiny compared with the main oxides. Still, “tiny” can change results fast. A few hundred ppm can decide whether the melt is calm or full of seeds. It can also decide whether a flint bottle looks water-clear or slightly green on one shift.
Trace Sb₂O₃ is most useful when the plant fights one of these:
- fast pull rate that leaves less fining time,
- variable cullet that swings oxidation demand,
- tight flint color windows,
- a forming/forehearth zone that tends to go slightly reducing and causes reboil.
Why redox control is a real business lever
Redox in glass is not abstract chemistry. It shows up as:
- Fe²⁺/Fe³⁺ balance and the visible tint of the bottle,
- sulfate behavior and foam,
- bubble growth or reboil in downstream zones,
- consistency of fining from day to day.
A variable-valence fining agent like antimony can act like a buffer. It does not replace good furnace control. It reduces the penalty when reality is not perfect. (See glass redox basics 1)
The simple mental model
| What the plant wants | What Sb chemistry can support | What can still break it |
|---|---|---|
| Fewer seeds | Oxygen release/uptake helps bubble growth and removal | Poor batch mixing, short residence time |
| Stable flint color | Redox buffering stabilizes Fe state | Cullet drift, burner imbalance |
| Less reboil | Redox control reduces oxygen “surprises” later | Forehearth reduction, carbon carryover |
| Faster ramp after changeover | More forgiving fining response | Wrong dosing, weak process control |
This is the main idea: Sb₂O₃ is often chosen when the plant needs more margin. The next sections answer the practical questions that usually come right after that.
The first question is the core one. What does Sb₂O₃ actually do inside the melt?
What roles does Sb₂O₃ play in melts?
Bubbles look simple. They are not. They are controlled by gas chemistry, viscosity, and how fast the melt can “feed” oxygen into existing seeds.
Sb₂O₃ plays two linked roles: it supports chemical fining through its Sb(III)/Sb(V) redox couple, and it acts as a redox buffer that stabilizes oxygen activity in the melt across temperature zones.

Role 1: chemical fining via oxygen release
Antimony can exist as Sb³⁺ and Sb⁵⁺ in silicate melts. That matters because a shift between these states can be tied to oxygen. In practical fining terms, oxygen released at the right time helps small bubbles grow. Bigger bubbles rise faster and sweep up smaller ones (Stokes’ Law). That is how chemical fining 2 “accelerates” bubble removal.
This is why antimony is often grouped with other variable-valence fining agents. In production language, it is an oxygen source or oxygen sink, depending on where the melt is in temperature and redox.
Role 2: redox buffering across the furnace → forehearth path
Many furnaces are not perfectly uniform. Hot spots, cold spots, and local fuel/air differences exist. A buffer does not eliminate those issues, but it reduces how much the melt response swings. With Sb present, oxygen activity can be steadier, which reduces sudden bubble events later. That steadiness is a major reason Sb has been historically “effective.”
Role 3: indirect support for sulfate fining behavior
Sulfate fining is common in mass glass. Sulfate needs the right oxidation environment to release SO₂/O₂ effectively at high temperature, while also avoiding excess foam. Redox balance impacts how much sulfate stays, how much is released, and how the melt behaves. Sb does not replace sulfate chemistry, but it can interact with the same redox landscape.
What this means for operators
| Melt issue | What Sb can help with | What Sb cannot fix |
|---|---|---|
| Persistent fine seeds | Boosts fining efficiency when tuned | Low temperature, short residence time |
| Reboil after fining | Stabilizes redox so late oxygen release drops | Mechanical air leaks, dirty cullet |
| Color drift in flint | Holds redox closer to target | Big Fe swings, wrong color package |
| Foam instability | Can reduce some redox-driven swings | Bad sulfate/carbon balance |
Sb₂O₃ is not a “magic clarifier.” It is a redox tool. It helps most when the furnace has normal small fluctuations, not when the process is out of control.
Now the next question is the one quality teams care about: how exactly does Sb improve fining and color consistency at the same time?
How does antimony improve fining and color consistency?
A flint bottle can pass dimensional checks and still fail the brand test. A slight green tint can kill a premium look. Seeds can kill filling yield.
Antimony improves fining by encouraging bubble growth and removal through redox-driven oxygen behavior. It improves color consistency because the same redox control stabilizes iron chemistry, which reduces shift-to-shift tint changes, especially in clear glass.

Fining: why fewer seeds also means fewer surprises downstream
In a typical furnace path, fining happens when the melt is hot enough and fluid enough for bubbles to grow and rise. If the melt later becomes slightly reducing, or if oxygen activity changes, bubbles can appear again. That is reboil.
Sb helps because it reduces the chance that the melt “changes its mind” as it travels. The goal is not only fewer bubbles at the end of the furnace. The goal is fewer bubbles at the feeder and in the gob.
Color: why redox is the hidden control knob
For many bottle colors, iron is the main driver. Fe²⁺ tends to push a green-blue tone. Fe³⁺ pushes yellow-brown. A stable ratio gives a stable look. When the furnace swings slightly reducing, more Fe²⁺ forms and the bottle looks greener. When it swings oxidizing, the tint shifts back. (See iron redox in glass 3)
Sb redox buffering can reduce these swings. It does not remove iron. It helps keep iron in the state the color package expects.
The “two birds” effect
Plants often see a paired outcome when Sb is tuned well:
- seeds and tiny bubbles fall,
- flint color variance tightens.
That is why Sb has been attractive in runs where both defects appear together.
Practical control signals that show Sb is doing its job
| Signal | What “better” looks like | What it usually means |
|---|---|---|
| Seed count trend | Lower average and lower variation | Fining is more stable |
| Forehearth reboil | Fewer late bubbles | Redox is not drifting late |
| Flint tint drift | Smaller ΔE / less green shift | Fe redox is steadier |
| Sulfate/foam behavior | Fewer foam events and fewer corrections | Redox balance is calmer |
When the plant sees these improvements, Sb dosing often gets credit. Still, this is the part that must be handled carefully: antimony is a regulated and watched substance in many markets.
So the next question becomes very practical. How much is “enough,” and how much becomes a risk?
What dosage keeps redox stable without regulatory flags?
Too little Sb does nothing. Too much makes people nervous. The best answer is not a single number. It is a controlled band tied to measurements.
A common “trace” approach keeps Sb₂O₃ at the lowest level that achieves seed and color targets, often in the range of a few hundred ppm up to around 0.05 wt% in the glass system when Sb is used as a fining aid. Compliance risk is managed through worker-exposure controls, customer heavy-metal policies, and migration/leach testing when required.
Dose for function, then lock the redox target
A plant should not dose Sb by habit. It should dose Sb against two measurable targets:
- seed rate / bubble count at the IS machine,
- redox proxy (often Fe²⁺/Fe_total, or an internal oxygen potential metric).
Sb works through valence chemistry. That means the best dosage depends on:
- sulfate and carbon levels,
- furnace atmosphere control,
- cullet oxidation demand,
- iron level in raw materials,
- pull rate and residence time.
So the “right” trace level is the smallest level that gives a stable redox signal and stable product outputs.
Avoiding regulatory and customer flags
Antimony trioxide is classified as a suspected carcinogen under EU CLP (see ECHA classification 4), and it is listed under California Proposition 65 for cancer warnings related to significant exposures. Those flags focus heavily on exposure pathways, especially inhalation risk for powders. That matters most in handling and batching, not in a finished glass bottle where Sb is bound in the glass matrix.
Still, customer programs often apply heavy-metal screens to packaging (like CONEG/TPCH 5). Some brand owners also apply conservative migration limits borrowed from plastic rules (see EU 10/2011 6). When the bottle is for food or pharma, the safe posture is simple: prove low release under relevant test conditions.
A practical “safe band” mindset
| Control item | Good practice | Why it reduces risk |
|---|---|---|
| Sb₂O₃ level | Use the minimum effective trace level | Lowers scrutiny and cost |
| Redox monitoring | Track Fe²⁺/Fe_total and color drift | Shows Sb is not “guesswork” |
| Batch handling | Enclosed transfer + dust control | Addresses main exposure concern |
| Product compliance | Run leach/migration checks when needed | Prevents surprises with big customers |
| Documentation | SDS + composition statement + test reports | Makes audits faster |
In many programs, the best answer is: keep Sb low, measure results, and document exposure controls. If a customer is sensitive to antimony as a “restricted substance,” then the cleanest solution is to move away from Sb entirely.
That leads to the final question: are antimony-free fining routes close enough to replace Sb for real bottle production?
Are antimony-free fining routes commercially viable soon?
Brands want lower-risk chemistry. Plants want stable quality and low cost. That tension is shaping the next wave of fining choices.
Antimony-free fining is already commercially viable in many mass bottle segments, because sulfate-based fining and modern furnace control can meet quality targets. For very tight seed specs or specialty applications, Sb-free routes like optimized sulfate systems, oxy-fuel/electric boosting, bubbling, and alternative fining agents (for example SnO₂ or CeO₂ systems) are progressing, but the timeline depends on product demands and furnace design.
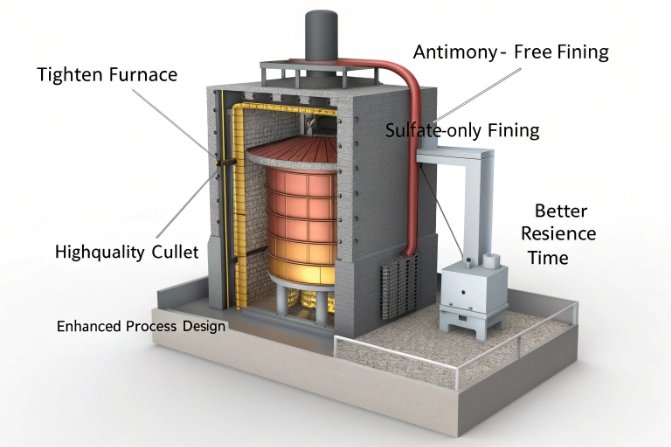
The reality today: most mass glass already moved away from Sb
For container and flat glass produced at huge scale, sulfur-based fining has been the main workhorse for a long time. The driver is cost, emissions management, and toxicology pressure. Sb and As (arsenic) were historically very effective, but they became harder to justify in many plants.
So “Sb-free soon?” depends on what “soon” means. For most beverage bottles, it is already here.
The harder cases: ultra-low seed flint, cosmetics, and high-clarity packs
Some segments still chase extremely low bubble counts and ultra-tight appearance. They also run more SKU changes and more cullet variability. Those cases are where Sb has stayed attractive.
The Sb-free toolbox for these cases often combines:
- better sulfate/carbon balancing,
- oxy-fuel or higher oxidation control (see oxy-fuel benefits 7),
- bubbling systems to speed bubble rise,
- better batch mixing and raw material sizing,
- electric boosting or local high-temperature zones,
- alternative fining chemistries (including tin- or cerium-based approaches in some designs). (See alternative fining agents 8)
“Commercially viable” depends on what you measure
| Segment | What counts as success | Sb-free outlook |
|---|---|---|
| Standard beverages | Low defects at high pull | Already viable at scale |
| Premium flint spirits | Tight tint + low seeds | Viable with strong control and trials |
| Cosmetics | Long shelf life + appearance | Viable, but needs strong compatibility testing |
| Pharma (special) | Very low risk + tight specs | Moving to higher-durability glass families and cleaner processes (see Type I glass 9) |
What is likely to change next
The strongest pressure is not only technical. It is supply chain policy. Retailers, regulators, and ESG programs (see glass sustainability 10) keep pushing for lower-risk additives and better worker safety. That pressure makes Sb-free improvements move faster, because plants invest in equipment and control systems that reduce the need for chemical “insurance.”
A realistic near-term outcome is:
- Sb use continues to shrink in mass container glass,
- Sb stays in a few niche quality corners unless those corners accept higher capex for Sb-free control,
- Sb-free options keep improving, so “niche corners” get smaller every year.
Conclusion
Sb₂O₃ is used as a redox tool that helps fining and color stability at trace levels. Still, Sb-free routes are already viable for many bottles and will keep expanding as process control improves.
Footnotes
-
Explains the concept of redox in glass melting and its impact on color. ↩
-
Overview of chemical and physical fining processes in glass manufacturing. ↩
-
Technical article on controlling iron redox to manage glass color. ↩
-
ECHA substance information on Antimony Trioxide classification. ↩
-
Information on Toxics in Packaging Clearinghouse (TPCH) heavy metal limits. ↩
-
EU Regulation 10/2011 on plastic materials intended to come into contact with food (migration limits). ↩
-
Benefits of oxy-fuel melting technology for glass furnaces. ↩
-
Research on alternative fining agents for glass melting efficiency. ↩
-
Data on Type I pharmaceutical glass and its high chemical durability. ↩
-
Sustainability initiatives in the European glass container industry. ↩








