Energy costs rise fast when a melt runs hot. Quality drops fast when a melt runs cold. Soda ash sits in the middle of that fight.
Soda ash (Na₂CO₃) is the main flux in most soda-lime container glass. It helps silica melt at practical furnace temperatures, but it must be balanced to protect durability, taste, and leach limits.

Why soda ash is the “speed lever” of container glass?
Soda ash turns silica into a workable melt
Pure silica melts too high for normal container furnaces. Soda ash breaks the silica network 1 during melting and forms sodium silicates 2. That drops the temperature needed to reach a workable viscosity. It also helps batch materials react faster, so the melt clears sooner and refines sooner.
In practical production, soda ash does not act alone. Limestone (or dolomite) brings CaO (and sometimes MgO) to stabilize the glass network that sodium weakens. Alumina helps too. This is the classic soda-lime balance: sodium helps melting, then lime and alumina help durability.
Soda ash is also a forming tool, not only a melting tool
Bottle forming needs a viscosity window 3 that is wide and stable. More sodium usually lowers viscosity at a given temperature. That can improve flow and mold fill, but it can also make the glass “too soft” if the line runs hot. Then defects rise, like thin walls, checks, and more scratches after anneal.
So the “right” soda level is not only about fuel. It is also about how the gob behaves in the mold.
Soda also shapes chemical risk and sensory risk
Sodium is the main alkali in standard container glass. Alkali makes glass more reactive with water-based foods over time, especially at high pH, high temperature, or long storage. If sodium is too high or the network is not stabilized well, leaching risk rises. For sensitive foods and drinks, that can show up as taste changes, haze, or label claim problems.
A simple way to explain soda ash to procurement
When buyers ask “why is the recipe changing,” a simple framework helps:
| What the brand wants | What soda ash helps | What soda ash can hurt | What the spec should control |
|---|---|---|---|
| Lower cost | Lower melt temperature | Higher leach risk if unbalanced | Alkali content window + leach test |
| Stable bottle weight | Better flow and forming | Thin-wall drift if too soft | Viscosity control + wall thickness |
| Long shelf life | Stable process gives stable glass | Hydrolytic durability may drop | Durability class + storage simulation |
| Better clarity | Faster melting and fining | More defects if batch is unstable | Seeds/bubbles limits + QC plan |
Soda ash is not a “cheap filler.” It is a control lever. When it is tuned well, energy drops and quality stays stable.
That sets the stage for the first key question: what exactly is Na₂CO₃ doing as a flux?
Soda ash looks like a raw material line item. In reality, it behaves like a chemical switch that changes the whole melt system.
What is Na₂CO₃’s function as a flux?
Batch reactions can stall. The sand does not fully react. The furnace runs harder. The bottle quality suffers. Soda ash exists to prevent that stall.
Na₂CO₃ acts as a flux by releasing sodium oxide into the melt. Sodium oxide reduces the melting and softening temperatures of silica-based glass and speeds up batch reactions.
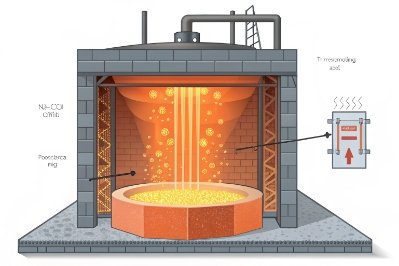
What happens in the furnace
Soda ash decomposes during heating. It releases CO₂ and leaves sodium oxide available to react with silica. This reaction forms sodium silicate phases that melt at much lower temperatures than silica alone. That early liquid phase is important. Once a liquid forms, it helps dissolve the remaining sand grains and pulls the batch toward a homogeneous melt.
This is why soda ash is called a flux. It “opens” the melt earlier and lets the furnace do more work with less peak heat.
Why container glass likes sodium more than other alkalis
Other alkalis can also lower melting temperature. Potassium compounds can do it. Lithium can do it even more strongly. But sodium is usually the best value and the most available at industrial scale. It also fits the well-known soda-lime network that container furnaces and forming lines are designed around.
In normal container glass, the goal is not the lowest possible melt temperature at any cost. The goal is stable melting, stable viscosity, stable durability, and stable cost. Sodium often hits that balance.
Flux power comes with a structure cost
Sodium breaks the silica network. That helps melting. But a “broken” network is less chemically durable unless it is stabilized. Lime and alumina are the stabilizers. If sodium goes up and stabilizers do not move with it, water resistance drops and alkali release can rise.
This is why flux design is always a recipe balance, not a single raw material decision.
| Item | What soda ash improves | What soda ash can worsen | Practical production signal |
|---|---|---|---|
| Melt temperature | Lowers required heat | Can increase corrosion load | Fuel rate trend |
| Batch reaction speed | Faster dissolution | More foam if unstable | Foam line behavior |
| Viscosity | Easier forming | Too soft gobs at same temp | Wall thickness drift |
| Durability | Indirect, if balanced well | Lower water resistance if unbalanced | Leach test drift |
| Clarity | Better fining potential | More bubbles if CO₂ release is poorly managed | Seed count trend |
A good flux plan also manages the gas release. CO₂ and other batch gases can create foam and seeds if fining and refining are not controlled. This is where furnace practice matters as much as chemistry.
Now it helps to connect sodium to energy in a direct way, since most buyers feel energy cost first.
Why sodium optimizes melt temperature and energy use?
Glass energy is not only about the flame. It is about how fast batch turns into clean melt and how long it stays in the “hard zone.”
Sodium lowers the working and melting temperatures of soda-lime glass, so furnaces can reach target viscosity with less heat input, less residence time, or both. That usually improves energy efficiency and throughput.

Lower temperature means more than lower fuel
When sodium content is optimized, the melt reaches homogeneity sooner. That reduces unmelted sand carryover and reduces defect risk. It can also allow a furnace to run at a lower crown or bath temperature for the same pull rate.
Lower operating temperature has a second benefit. It can reduce volatilization and reduce some types of refractory wear 4. But this depends on the total batch chemistry and the furnace atmosphere. Sodium itself can increase alkali vapor and can attack certain refractories if the furnace is not designed well. So “lower temp” does not always mean “lower wear.” It means there is a new balance to manage.
Sodium supports cullet use, and cullet saves energy
Cullet is already glass, so it melts faster than raw batch. Higher cullet ratios 5 can lower energy per ton and can improve melt stability. Sodium helps the whole batch system, but cullet is often the biggest energy lever available to container plants.
The best results usually come from pairing:
- a stable sodium window
- high, clean cullet percentage
- steady batch charging and good mixing
Energy saving must not create quality drift
Energy saving is not useful if it increases rejects. If sodium is pushed too high to chase lower temperature, viscosity can drop too much. Then the forming line needs lower temperatures or slower speeds. That can erase the furnace savings.
The correct way to optimize energy is to choose a sodium range that supports the forming window and then improve energy through furnace design and cullet strategy.
| Energy lever | Role of sodium | What to watch | What to record in audits |
|---|---|---|---|
| Lower melt setpoint | Makes it feasible | seeds, cords, viscosity drift | furnace temp, defect counts |
| Higher pull rate | Helps batch melt faster | fining limits | bubbles/seed trend |
| More cullet | Works well with stable recipe | contamination, color drift | cullet % and sorting method |
| Electric boosting | Sodium still needed for melt chemistry | electrode corrosion risks | power input vs pull rate |
| Oxy-fuel / heat recovery | Sodium window can be tightened | NOx, alkali carryover | emissions and crown deposits |
Sodium is the chemical part of energy control. Furnace technology is the mechanical part. Both must work together.
Next comes the harder trade-off question: how to keep sodium benefits while staying inside durability and leach limits.
How to balance soda with durability and leach limits?
A bottle can be cheap and clear, then still fail in the market because the product taste shifts. This is where durability specs matter.
Balancing soda means setting a controlled Na₂O window and matching it with stabilizers like CaO and Al₂O₃, plus process controls that protect the inner surface. Durability must be proven with leach testing and storage simulations for the real food.
Understand what “leaching” looks like in foods
Leaching is not always dramatic. For many foods, it is a slow exchange at the glass surface. The first stage is often alkali release and ion exchange with water-based fills. Over time, the inner surface can change. In some products, this can show up as:
- slight taste shift
- haze or sediment
- color instability
- faster oxidation due to surface effects in special formulas
Not every product is sensitive. But acidic foods, high-temperature hot fill, long storage, and sensitive botanicals push risk up.
Recipe balance is the first control
In soda-lime glass, sodium provides workability. Lime and alumina add durability. Magnesium can help stability too. A practical approach is to control sodium tightly and avoid “hidden” sodium increases from raw material variability and cullet contamination.
If a buyer needs high hydrolytic resistance 6, it is not enough to say “soda-lime.” The spec must call out performance. That performance is proven by standardized durability tests and by product-specific storage tests.
Surface quality and process stress also matter
Even with a good recipe, surface attack can rise if the bottle inner wall is rough, micro-cracked, or contaminated. Process steps like aggressive washing, strong caustic contact, or poor annealing can raise risk. For sensitive foods, the safest plan is a combined approach:
- recipe control
- anneal stress control
- clean, stable inner surface
- optional barrier layers when needed
Write the balance into procurement language
A procurement spec should not try to control every oxide. It should control what matters for risk: a chemistry window, a durability outcome, and a method to verify it.
| Spec area | What to set | How to verify | Why it prevents disputes |
|---|---|---|---|
| Chemistry | Na₂O range and key stabilizers range | batch records + periodic lab analysis | stops drift |
| Durability | hydrolytic resistance target | standard durability test + trend | ties to performance |
| Leach behavior | product-specific limit | fill-and-hold simulation | matches real risk |
| Inner surface | defect and cleanliness limits | visual + rinse + particle checks | reduces variability |
| Change control | cullet source and raw material changes | supplier notification rule | avoids silent shifts |
For many food and beverage brands, the most practical approach is to set UV-VIS transmittance specs and durability specs side by side. Light control protects flavor. Durability control protects taste and clarity over time.
Now the last question is strategic: can low-soda strategies work when furnaces get better?
Are low-soda strategies viable with advanced furnaces?
Some buyers want lower alkali for better durability. Some plants want lower alkali vapor for cleaner furnaces. The question is whether modern furnace tech can carry the melt without “classic” soda levels.
Low-soda strategies are viable in limited ranges when furnace efficiency is high and cullet use is strong. Still, sodium remains the core flux for most container glass, so “low-soda” usually means “optimized soda,” not “no soda.”

What “low-soda” really means in container glass
In most container plants, removing too much sodium forces higher temperatures or longer residence time. That raises energy and can raise defects. So low-soda is usually a modest reduction paired with stronger stabilizers and better melting practice.
The goal is often one of these:
- improved chemical durability for sensitive fills
- reduced alkali volatilization 7 and deposits
- improved compatibility with specific coatings or decorations
Advanced furnaces help, but they do not change chemistry limits
Modern tools can make lower-soda recipes more practical:
- higher cullet ratios with good sorting
- electric boosting for more uniform heat in the melt
- oxy-fuel combustion and better heat recovery
- batch preheating and better charging control
- improved mixing and fining control
These tools reduce the penalty of lower sodium, because the furnace can supply heat more efficiently and keep the melt more uniform. But the chemistry still matters. Silica still needs flux to melt at container scale.
Alternative flux paths exist, but they carry cost and risk
Some plants explore potassium or lithium compounds. These can lower melting temperature, but they are often more expensive and can change viscosity behavior and durability. They can also change color response and volatilization behavior. For large-scale bottles, the simplest path is still sodium-based flux with better controls.
A more realistic “low-soda” path is to keep sodium in a stable range, raise cullet, and tune lime and alumina for durability, while using furnace upgrades to hold energy down.
| Low-soda approach | What makes it work | Main risk | Best use case |
|---|---|---|---|
| Modest Na₂O reduction + more stabilizers | strong fining and stable forming | viscosity shift | sensitive foods needing better durability |
| Higher cullet ratio | clean cullet stream | contamination and color drift | high-volume clear bottles |
| Electric boosting | uniform melt heat | electrode wear, capital cost | plants needing tighter control |
| Oxy-fuel / heat recovery | higher thermal efficiency | deposit patterns change | energy-sensitive regions |
| Alternative alkali flux | stronger flux power | cost and recipe complexity | niche high-value bottles |
Low-soda strategies can work when the whole system is ready. The best projects treat recipe, furnace, and forming as one plan.
Conclusion
Soda ash is the key flux that makes container glass practical. The winning recipe keeps sodium optimized for energy and forming, then locks durability with stabilizers and verified leach performance.
Footnotes
-
Learn how the chemical arrangement of the silica network determines the melting point and stability of glass. ↩ ↩
-
A technical guide to sodium silicates and their function as vital intermediates during the glass melting process. ↩ ↩
-
Understanding the viscosity window is essential for controlling the flow and cooling rate during automated bottle forming. ↩ ↩
-
Detailed analysis of how high-temperature chemistry and alkali vapors contribute to the gradual refractory wear of furnace linings. ↩ ↩
-
Insights into how optimizing cullet ratios reduces energy demand and extends the operational lifespan of glass furnaces. ↩ ↩
-
A guide to testing hydrolytic resistance to ensure glass packaging meets safety standards for food and medicine. ↩ ↩
-
Research regarding alkali volatilization and its effects on glass quality and industrial furnace emissions control. ↩ ↩








