Too much lime looks like a quick durability fix. Then haze, stones, and random cracks appear, and the whole line turns into firefighting.
For standard soda-lime bottle glass, CaO is usually kept around 8–12 wt%. Many container plants treat ~12 wt% as a practical upper ceiling for flint bottles, unless the full Na₂O–CaO–MgO balance and liquidus margin are proven stable on the furnace.

CaO “upper limit” means a practical ceiling, not one universal number?
Typical container-glass CaO bands
In most soda-lime container recipes 1, CaO lives in a tight band because it is doing a specific job: it stabilizes the network that Na₂O opens. If CaO is too low, durability drops and the glass can feel “soft” in water or humid storage. If CaO is too high, crystallization risk rises and the melt becomes less forgiving in cooler zones.
That is why I prefer two definitions:
-
Practical upper limit (operations limit): the highest CaO that still runs clean on the real furnace with low haze, low stones, stable color, and stable forming.
-
Absolute upper limit (chemistry limit): the point where the liquidus and devit behavior become unsafe for normal container forming, even if the melt looks okay during a short trial.
For many flint bottle lines, the practical ceiling ends up near the top of the common 8–12 wt% band. Some furnaces can run slightly above that, but only with strong control of MgO, Na₂O, fining chemistry 2, and forehearth temperature uniformity.
What really sets the ceiling in a bottle plant
CaO is not capped because “chemists like 10%.” It is capped because bottles are made in a long thermal path:
-
hot furnace
-
refining zone
-
forehearth
-
feeder
-
forming molds
-
annealing lehr
Crystals do not need to form in the hottest zone to cause problems. They often start in cooler or mixed-temperature zones, then show up as haze, stones, or local stress points.
A simple “CaO ceiling” decision table
| If CaO goes up… | Benefit you might get | Risk you might trigger | What I require before approving |
|---|---|---|---|
| +0.2 to +0.5 wt% | small durability gain | higher liquidus margin pressure | stable seed/stones trend for a full campaign segment |
| +0.5 to +1.0 wt% | stronger chemical stability | devit haze in forehearth | haze/stone counts + glass chemistry control charts |
| >+1.0 wt% | niche tuning only | cords, crystals, unstable forming | liquidus modeling + controlled trial + strict stop rules |
The procurement-friendly way to state an “upper limit”
Instead of one hard number, I set:
-
a target CaO range for the SKU family
-
a hard cap that triggers re-qualification
-
and process KPIs that prove devit margin (haze, stones, cords, seed counts)
This keeps buyer–supplier discussions objective. It also prevents the “same COA, different behavior” problem that shows up when cullet shifts.
The next step is to explain the real constraints that decide maximum calcium content. Once those constraints are visible, CaO decisions become much easier.
What constraints set maximum calcium content?
A CaO number looks simple on a certificate. In a furnace, it is linked to temperature, residence time, and the way crystals try to form in cooler zones.
Maximum CaO is constrained by liquidus temperature, devitrification margin in the forehearth, viscosity/working range, and the durability target that the bottle must meet. The tightest constraint usually wins.

Liquidus temperature and devit margin
The most important technical constraint is liquidus behavior 3. If the liquidus is too close to the forehearth or feeder temperatures, crystals can form. Even a small amount can cause:
A bottle plant does not need “zero crystals forever.” It needs enough margin that crystals do not form under normal drift.
Viscosity and the forming window
CaO also affects viscosity behavior 5 through the full oxide balance. If a CaO increase forces higher temperatures to hold forming viscosity, the plant may see:
-
higher volatility and deposits
-
more correction cycles
-
higher color drift risk (redox sensitivity)
In practice, the upper CaO limit is often where the plant can no longer keep a stable gob without pushing heat.
Durability class and customer expectations
CaO supports durability, but it is not the only lever. If a project demands tighter hydrolytic resistance 6, it can be safer to build margin using:
-
balanced MgO
-
modest Al₂O₃
-
cleaner cullet streams
instead of pushing CaO alone.
Constraint map table
| Constraint | “Too high CaO” symptom | Where it appears first | How to measure it fast |
|---|---|---|---|
| Liquidus/devit margin | haze, stones, devit streaks | forehearth 7 / feeder | haze inspection + stone counts + chemistry trend |
| Working range | unstable gob, thickness spread | feeder / forming | thickness map + reject rate by defect type |
| Color/redox stability | tint drift, more corrections | furnace control | ΔE trend + redox proxies + correction logs |
| Durability target | tempting to push CaO | long-term storage | durability screening + field feedback |
How I translate this into a “max CaO” rule
-
If devit risk is already present, CaO is capped lower, and MgO/Al₂O₃ are used for stability.
-
If devit risk is low and the furnace is stable, CaO can sit near the top of the normal band, but only with tight monitoring.
Those constraints explain why “upper limit” varies by furnace and SKU. The next question is the big why: why does high CaO raise crystallization risk so quickly?
Why too much CaO raises crystallization risk?
Crystallization does not start because someone “added too much lime.” It starts because the melt becomes closer to calcium-silicate crystal fields as it cools through sensitive temperatures.
Too much CaO raises crystallization risk because it pushes the composition toward calcium-silicate and sodium-calcium-silicate phases. As the glass cools in the forehearth, these phases can nucleate and grow, creating haze, stones, and optical distortion.
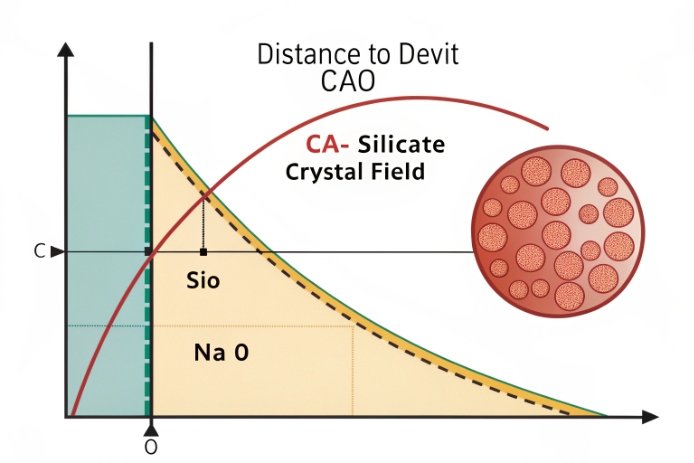
What devitrification looks like in bottle reality
In soda-lime container glass, common devit products discussed in practice include:
-
wollastonite-type calcium silicates
-
devitrite-type sodium–calcium silicates
The exact phase mix depends on the full Na₂O–CaO–SiO₂ balance, plus minor oxides and thermal history.
Crystals can start small and invisible, then become visible as:
-
a cloudy veil in thick zones
-
bright “sparkle” points
-
refractive streaks that distort label appearance
Why forehearth temperature drift becomes dangerous
A furnace can look fine at the melt surface and still devit in the forehearth. The forehearth is where:
-
temperature is lower
-
gradients exist
-
residence time can be long enough for growth
A CaO increase can reduce the safety margin so much that a normal seasonal drift becomes a devit event.
Why “more CaO for durability” can backfire
When crystals form, durability can look worse, not better:
-
crystals create interfaces and stress points
-
optical defects rise
-
bottle strength scatter increases
So a durability-driven CaO push can end up reducing total quality.
Devit risk table (simple and practical)
| Driver | What shifts with higher CaO | What defect shows up | What I do instead of “more CaO” |
|---|---|---|---|
| Liquidus rises | less margin to feeder temps | haze, stones | adjust CaO/MgO split, stabilize temps |
| Crystal fields expand | easier nucleation | cords/striae-like distortion | tighten batch mixing + reduce peaks |
| Thick-base optics | longer path length | base looks cloudy | keep CaO stable, tune design and cooling |
The key lesson
Crystallization risk is not only about an average CaO number. It is about how close the plant runs to crystal growth conditions. That is why CaO is usually capped in bottle recipes, even when durability targets are aggressive.
Now the practical question: if CaO is capped, how can durability still be met?
How to cap CaO while meeting durability targets?
Many buyers want “stronger chemical resistance.” CaO helps, but it is not the only lever, and it is not always the safest lever at the top end.
Meet durability targets without pushing CaO by balancing CaO with MgO, using modest Al₂O₃ for network stability, holding Na₂O stable, and tightening cullet and melting control. The goal is a stable surface and low defect rate, not a single high CaO number.
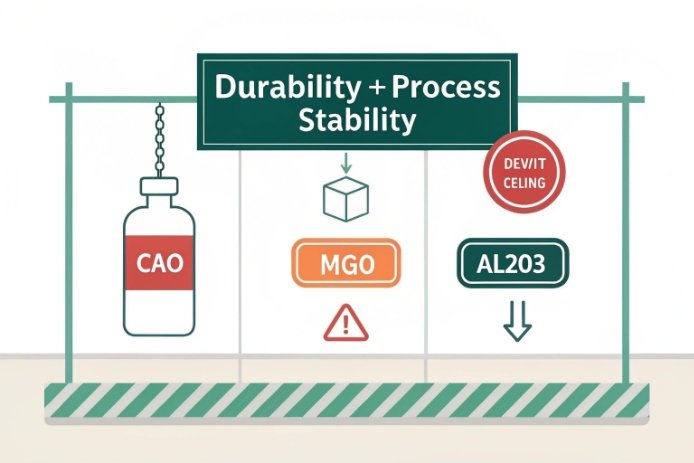
Use the “stability package” instead of one oxide
A practical durability package in soda-lime bottles often looks like:
-
CaO in a controlled band (not at the cliff edge)
-
MgO tuned to support durability and reduce devit tendency
-
Al₂O₃ used modestly to tighten the network and reduce leaching
-
Na₂O held stable so the surface does not become too mobile
This approach improves durability while keeping the melt and forehearth safer.
Fix the surface reality, not only the chemistry
Durability complaints often come from surface condition:
-
scuffs and micro-scratches
-
residual stress
-
humid storage reactions
So durability programs should also include:
A durability-first tuning table
| Durability goal | Safer lever than “higher CaO” | Why it works | What to validate |
|---|---|---|---|
| Lower alkali leaching | modest Al₂O₃ increase | tighter network | durability screen + viscosity guardrail |
| Better humidity stability | MgO balance + clean cullet | less drift, steadier surface | MgO trend + haze/seed trend |
| Returnable/caustic wash | surface protection + stable stabilizers | fewer flaw starters | wash cycling + scuff KPI |
| Long shelf-life foods | stable chemistry + low defects | fewer weak points | field storage test + optical checks |
How I write the spec so it stays enforceable
-
Set CaO range and do not exceed cap.
-
Add a CaO/MgO balance statement if the SKU is devit sensitive.
-
Require durability proof by a defined screening method.
-
Add a “cullet stream change triggers re-qualification” clause.
This keeps durability gains real and repeatable, not a one-time lab win.
The last question matters more each year: recycled cullet is rising, and that changes how “fixed limits” work in practice.
Are adaptive limits used with high-cullet feeds?
High recycled cullet 9 is good for carbon and cost. It can be hard on chemistry stability if the cullet stream is not controlled.
Yes. Many plants use adaptive control limits for CaO when cullet varies, but they do it with guard bands and alarms. The target stays tight, while short-term limits flex based on measured cullet chemistry and predicted devit margin.

Why cullet pushes CaO variability
Cullet brings in the chemistry of many container families. If the stream is not closed-loop, CaO and MgO can drift because:
-
different bottle families use different alkaline-earth balances
-
fines and contamination change melting and redox behavior
-
color sorting errors trigger more correction cycles
So a fixed CaO number may be “right,” but the incoming mix may not let the plant hold it without constant batch corrections.
What “adaptive limits” really look like
Adaptive limits are not “we accept anything.” They are a structured plan:
-
Target range stays narrow for the SKU.
-
Short-term operating range can move slightly when cullet chemistry shifts.
-
Hard cap stays in place to prevent devit risk.
-
Re-qualification trigger fires if the plant needs to run near the cap too often.
A simple adaptive-control table
| Control layer | Example rule | Why it helps | What it prevents |
|---|---|---|---|
| Target CaO range | tight band for the SKU | keeps product stable | buyer disputes |
| Guard band | early warning near the cap | gives time to correct | devit surprises |
| Hard cap | stop-and-review threshold | protects forehearth margin | haze and stones |
| Cullet chemistry gate | reject or regrade cullet lots | stabilizes inputs | constant batch correction loops |
| Trending & SPC 10 | weekly CaO/MgO charts | catches drift early | “sudden” quality drops |
How to make adaptive limits buyer-safe
If a buyer is worried about variability, the contract can require:
-
trending reports for CaO and MgO
-
cullet grade definitions (flint-only, contamination limits)
-
a change-control notice when cullet sources change
-
proof that devit and optical KPIs stayed within limits
Adaptive limits work best when the plant treats cullet like a controlled raw material, not a free melting aid.
Conclusion
For most soda-lime bottles, CaO is usually capped near the top of the common 8–12 wt% band to avoid devitrification. Durability gains should come from balanced CaO/MgO, modest Al₂O₃, and tight process control.
Footnotes
-
The most common commercial glass type, composed mainly of silica, soda, and lime for container manufacturing. [↩] ↩
-
The chemical and physical process of removing gas bubbles from molten glass to ensure clarity. [↩] ↩
-
The temperature above which a material is completely liquid; cooling below this point risks crystallization. [↩] ↩
-
Solid inclusions in glass caused by unmelted batch or refractory materials, creating stress. [↩] ↩
-
A graphical representation of how glass fluidity changes with temperature, critical for controlling forming stability. [↩] ↩
-
A measure of glass durability that defines its resistance to releasing elements when in contact with water. [↩] ↩
-
A refractory channel that conveys molten glass from the furnace to the forming machine while conditioning temperature. [↩] ↩
-
A controlled cooling process designed to relieve internal residual stresses in formed glass containers. [↩] ↩
-
Broken or waste glass re-melted to lower energy consumption and raw material costs in production. [↩] ↩
-
A method of quality control using statistical methods to monitor and control the glass manufacturing process. [↩] ↩








