Leachables problems rarely appear on day one. They appear after stability testing, then the whole launch plan gets stuck.
Glass extractables and leachables are driven by alkali content, network strength, surface condition, and process stability, so the formulation and furnace controls decide pH rise and ion release.

Why does formulation control extractables and leachables in glass bottles?
The simplest truth is that water attacks the glass surface first. It does not “dissolve the bottle.” It swaps ions, builds a hydrated surface layer, and slowly releases a small set of elements into the liquid. That release can change pH, can add ionic impurities, and can trigger drug instability in sensitive formulations.
Two mechanisms explain most of what is measured in hydrolytic resistance and in extractables and leachables (E&L) screening 1.
Ion exchange is the pH-rise engine
When water contacts soda-lime or alkali-containing glass, H⁺ (or H₃O⁺) exchanges with Na⁺ and K⁺ at the surface. Na⁺ enters the liquid, H⁺ enters the glass surface, and the solution becomes more basic. That is why higher Na₂O often means a higher pH rise in water extraction. The exchange is faster when the surface is rough, stressed, or chemically inhomogeneous.
Network dissolution is the “deeper” release pathway
After ion exchange, the hydrated layer can allow slower network dissolution. This is where Si, B, Al, Ca, and Mg may appear, depending on glass type and test severity. A tighter network (higher SiO₂ and modest Al₂O₃) tends to slow this pathway. A surface with micro-defects, devit skins, or cords tends to speed it up.
Why the same recipe can show different results
Extractables are surface-driven. The surface can change even when bulk composition is stable. Cullet contamination, sulfate fining swings, redox swings, and cooling profile changes can create local defects that increase effective surface area and add weak zones. This is why E&L control is never “composition only.”
| Lever | What it changes | What shows up in tests |
|---|---|---|
| Na₂O / K₂O level | Ion-exchange capacity | Faster pH rise and higher alkali release |
| SiO₂ + Al₂O₃ strength | Network durability | Lower total ionic release over time |
| CaO/MgO balance | Stability vs devit risk | Indirect effect through surface quality |
| Impurities (Fe, Cr, Ni, heavy metals) | Trace leachables and defect risk | Metals in extract + more defects |
| Surface treatment / coating | Surface alkali availability | Lower surface hydrolytic extractables |
The best results come from a clear plan: choose a glass family that can meet the hydrolytic class, then control surface condition so the test reads the glass, not the defects.
Keep reading, because the next sections turn this into specific oxide choices, composition targets, and daily production controls.
Which oxides and impurities (Na₂O, CaO/MgO, Al₂O₃, B₂O₃, Fe₂O₃, heavy metals) most influence pH rise and ionic leaching?
High E&L results often start with one mistake: chasing melting ease with high alkali, then trying to “test around” it later.
Na₂O (and K₂O) most directly drives pH rise and early alkali release, while SiO₂ and modest Al₂O₃ slow long-term ion release; CaO/MgO mainly matter through stability and surface quality, and heavy metals matter as trace leachables and defect starters.
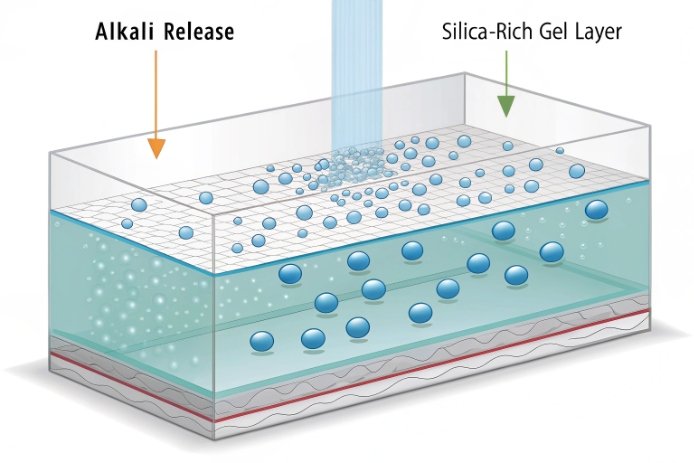
Na₂O and K₂O: the main pH lever
If the goal is low pH rise in water extraction, alkali level is the first place to look. Na₂O increases melt workability, but it also increases the pool of exchangeable Na⁺ at the surface. In hydrolytic tests that measure released alkali by titration, Na₂O content and surface condition dominate the result. K₂O behaves similarly, even if its absolute level is usually lower in container glass.
Al₂O₃ and SiO₂: the “durability backbone”
Al₂O₃ strengthens the glass network and often improves chemical durability. In many container and pharma glass systems, a modest Al₂O₃ increase reduces the rate of network dissolution after ion exchange. SiO₂ is the main backbone. Higher SiO₂ generally supports durability, but it can raise melting temperature and energy demand. The best strategy is to hold SiO₂ in a stable window and use Al₂O₃ as a controlled durability tool.
B₂O₃: helpful in Type I families, sensitive in process
B₂O₃ is central in many borosilicate Type I glasses. It supports high hydrolytic resistance by forming a durable network with SiO₂ and Al₂O₃, while keeping viscosity workable. Still, B₂O₃ can introduce boron as a measurable extractable element in some E&L screens, so the acceptance criteria must match the formulation risk.
CaO and MgO: indirect drivers through surface and devit
CaO and MgO stabilize soda-lime glass. They can reduce alkali mobility when balanced well, but their biggest E&L impact is indirect. Poor CaO/MgO balance can raise devit risk, and devit skins increase surface roughness and leaching. So CaO/MgO is a “surface quality control” lever more than a pH lever.
Fe₂O₃ and heavy metals: small concentration, high attention
Iron is not usually the main cause of pH rise. It matters when it travels with cullet contamination, redox instability, and cords. Heavy metals (Pb, Cd, As, Sb, Cr, Ni, Co) are controlled because they can show up as trace leachables and can raise regulatory risk. Even when present at low levels, they must be managed with raw material and cullet specs and aligned to frameworks such as the ICH Q3D(R2) elemental impurities guideline 2.
| Oxide / impurity | Main E&L effect | Practical control action |
|---|---|---|
| Na₂O / K₂O | Faster ion exchange → pH rise | Keep alkali in a tight window, reduce drift |
| SiO₂ | Slower network dissolution | Avoid “low silica” shortcuts for melting |
| Al₂O₃ | Higher durability, slower leaching | Target modest increase (often ~1–2% in many systems) |
| B₂O₃ | Type I durability, boron release risk | Match glass family to drug sensitivity |
| CaO/MgO | Surface quality via devit behavior | Balance ratio, avoid cold-zone devit |
| Heavy metals | Trace leachables + defect risk | Tight COA limits, strict cullet governance |
The fastest win is often not a new recipe. It is tighter alkali control, tighter impurity limits, and a cleaner, more stable surface coming out of the hot end.
What composition targets help pass USP <660>, EP 3.2.1, and ISO 4802 hydrolytic resistance tests?
Many teams treat compendial tests like a paperwork step. In reality, these tests define what glass family and what surface condition is acceptable for pharma use.
Passing USP <660>, EP 3.2.1, and ISO 4802 usually means selecting a Type I (borosilicate/aluminosilicate) glass for highest hydrolytic resistance, or using Type II treated soda-lime when the product and regulation allow, then holding composition and surface condition stable so hydrolytic class does not drift.

In practice, teams anchor requirements to USP <660> Containers—Glass 3, the European Pharmacopoeia general chapter 3.2.1 4, and ISO 4802 hydrolytic resistance methods 5—then work backward to a glass family and surface strategy that stays stable across lots.
Start by matching glass type to risk: Type I vs Type II vs Type III
For most high-risk parenterals and sensitive aqueous drugs, Type I is the safest baseline because the bulk composition itself is highly hydrolytically resistant. Type II is a soda-lime base with an inner surface treatment that improves hydrolytic resistance. Type III is untreated soda-lime with moderate resistance and is used when product risk is lower.
This is where composition targets become practical:
- If the target is Type I performance, alkali must be low and network formers must be stronger.
- If the target is Type II, the base soda-lime must be consistent and the inner surface treatment must be stable.
- If the target is Type III, the recipe can be standard soda-lime, but E&L expectations must be realistic.
Practical composition direction by glass family
Instead of promising one universal oxide recipe, I prefer “directional targets” that procurement and production can hold.
- Type I borosilicate direction: higher SiO₂, meaningful B₂O₃, meaningful Al₂O₃, lower total alkali, controlled alkaline earths.
- Type I aluminosilicate direction: higher SiO₂ and higher Al₂O₃, lower alkali, and lower or no B₂O₃ depending on supplier design.
- Type II/III soda-lime direction: standard soda-lime base with controlled Na₂O and tighter impurity control, plus (for Type II) inner surface dealkalization.
Why “targets” must be tied to surface tests
USP and ISO methods for surface hydrolytic resistance attack the inner surface with hot water and then quantify alkali release. These tests punish rough surfaces, devit skins, and inhomogeneity. So the target is not only oxide %. It is “oxide % + surface condition.”
| Glass type goal | Composition direction | Surface strategy | Typical risk it mitigates |
|---|---|---|---|
| Type I performance | Lower alkali, stronger network (SiO₂/Al₂O₃/B₂O₃ family) | Keep surface clean, low defects | Low alkali release, stable pH |
| Type II performance | Standard soda-lime held stable | Inner surface dealkalization | Lower surface alkali release |
| Type III performance | Standard soda-lime | Good melt quality and cleanliness | Moderate durability for lower-risk products |
A buyer-friendly way to specify this
A specification that avoids disputes includes:
- glass type intent (Type I / II / III)
- hydrolytic class acceptance based on the relevant standard
- composition limits for alkali and heavy metals
- process limits that protect surface quality (devit, cords, stones)
When this is written clearly, compendial compliance becomes predictable, and E&L becomes a controlled outcome rather than a surprise.
How do cullet percentage, furnace redox, and fining chemistry change alkali release in water/acid simulants?
When E&L numbers drift across lots, the recipe is usually blamed first. Most of the time, the drift starts with cullet and process stability.
Cullet can raise alkali and impurity variability, redox shifts can change sulfate behavior and surface homogeneity, and fining chemistry can leave different salt residues and defect populations, so all three can move alkali release in water and acid simulants even with the same nominal composition.

Cullet percentage: melt helper, chemistry noise source
Cullet usually improves melting and can reduce unmelted batch grains, which can help surface quality. Still, cullet also brings unknowns:
- higher or drifting Na₂O and CaO depending on source
- ceramics, stones, and metals that create micro-defects
- trace metals that become measurable leachables
- organics that change the local redox near the doghouse
For E&L stability, the key is not only the cullet ratio. The key is cullet consistency and cleanliness. A stable cullet stream at 50% is often safer than an unstable stream at 30%.
Furnace redox: a hidden lever for surface uniformity
Redox changes do not directly “add sodium,” but they change melt behavior. Redox swings can:
- shift the balance of fining reactions
- change sulfate retention and foam behavior
- increase cords and striae, which can reach the surface
- create local weak zones where water attacks faster
If hydrolytic results move with day-night furnace swings, the root cause is often combustion and pull stability, not bulk chemistry.
Fining chemistry: defect population affects apparent leaching
Hydrolytic tests and E&L screens are sensitive to surface flaws. Poor fining leaves more seeds and can create surface pits. Sulfate fining swings can change salt carryover behavior and can create local inhomogeneity. Some fining additives can also introduce trace elements that appear in E&L profiles.
Water vs acid simulants: why the trend can change
In water, alkali release and pH rise are often dominant. In acid simulants, ion exchange can still occur, but the chemistry often favors different release patterns, and alkaline earth ions can become more visible depending on conditions. This is why E&L programs should define the simulant set clearly and link it to product chemistry.
| Variable | What it changes | What it can do to E&L numbers | Best daily control |
|---|---|---|---|
| Cullet % and quality | Alkali and impurity inputs | Lot-to-lot drift in alkali and trace metals | Incoming cullet QC + blending plan |
| Redox stability | Fining and homogenization | More cords/defects → higher apparent leaching | Stable oxygen potential and pull |
| Fining agent stability | Seeds, salts, surface pits | Higher extraction due to defects | Stable fining feed and fining temperature |
| Cooling profile | Surface stress and hydration risk | Faster attack on weak surface zones | Stable forehearth and annealing control |
A stable melt produces a stable surface. A stable surface produces stable extractables. That is the clean chain that makes audits easier.
Can hot-end sulfur treatment or internal SiO₂ coatings cut extractables without altering the base recipe?
Sometimes the base recipe is locked by cost, furnace design, or supply chain. In that case, surface engineering becomes the practical path.
Yes. Hot-end sulfur treatment (Type II dealkalization) can reduce surface alkali release by modifying the inner surface, and internal SiO₂ barrier coatings can sharply reduce extractables, but both must be qualified because they change the surface that the compendial tests measure.

Hot-end sulfur treatment: improving surface hydrolytic resistance
Type II glass commonly begins as soda-lime and then receives an inner surface treatment. A sulfur-based hot-end treatment exposes the hot glass surface to sulfur-containing gases, which can dealkalize the surface and create a more durable, silica-rich layer. This reduces the amount of alkali that can exchange into water under the hydrolytic surface test conditions.
This approach can be attractive because it does not require changing the bulk glass recipe. Still, it requires strict process control:
- stable treatment conditions
- stable container temperature profile
- verification that the treatment effect remains consistent across shifts
If treatment is uneven, E&L results can become more variable, not less.
Internal SiO₂ coatings: a strong barrier, stronger qualification burden
Internal SiO₂ coatings can act as a true barrier layer between the product and the bulk glass. In many cases, this sharply reduces alkali and trace metal extractables. It is a powerful tool for sensitive drugs, especially when a “clear” bottle look is required.
The trade-offs are qualification and testing scope:
- coatings can change surface test applicability and interpretation
- coating integrity must be verified (coverage, defects, adhesion)
- fill-finish compatibility must be confirmed
How to choose between treatment and coating
The practical selection depends on product risk and cost tolerance.
| Option | Best for | What it reduces | Main risk |
|---|---|---|---|
| Hot-end sulfur treatment (Type II) | Many aqueous pharma products where soda-lime is acceptable | Surface alkali release | Drift if treatment is not uniform |
| Internal SiO₂ coating | High-risk formulations, very low extractables targets | Broad reduction in extractables | Coating qualification and integrity control |
| Base recipe change (Type I glass) | Highest hydrolytic resistance baseline | Bulk-driven durability | Cost, supply chain, forming changes |
A surface solution should never be treated as a shortcut. It must be validated like a material change, because it changes the contact surface that defines extractables behavior.
Conclusion
Extractables and leachables are controlled by alkali level, network strength, impurities, and surface quality, so the best results come from stable composition plus stable cullet, redox, and surface treatment discipline.
Footnotes
-
FDA draft Q3E outlines risk-based approaches to assess and control E&L in packaging systems. (↩︎) ↩
-
ICH Q3D(R2) provides permitted daily exposures and a risk-based control strategy for elemental impurities. (↩︎) ↩
-
USP briefing explains intent and performance-based approach behind chapter <660> for glass containers. (↩︎) ↩
-
EDQM notice summarizes updates to Ph. Eur. 3.2.1 tests defining glass types and hydrolytic resistance. (↩︎) ↩
-
ISO entry describes ISO 4802 test principles for measuring alkali release from internal glass surfaces. (↩︎) ↩








