Scratches look small, but they can ruin shelf appeal and trigger cracks on the line. Many bottle failures start with one hidden abrasion mark.
Alumina (Al₂O₃) can improve scratch and abrasion resistance because it strengthens the glass network and stabilizes the surface. The benefit is real, but it must be balanced with melting temperature, viscosity, and devitrification risk.

The real relationship between Al₂O₃ and “scratch resistance”
Scratch resistance 1 in bottles is not only “hardness.” A bottle scratches when the surface is damaged by contact, and that damage can turn into micro-cracks. Those micro-cracks then grow under stress, moisture, and impact. Alumina helps mainly because it makes the glass network tighter and makes the surface more stable against water-driven weakening. That matters on filling lines where bottles slide, rub, and tap each other thousands of times per hour.
It is also important to be honest: alumina is not a magic shield. Most abrasion on filling lines is controlled by surface condition, coatings, handling, and line lubrication. Chemistry sets the baseline. Process decides the final result.
What scratch resistance really includes
-
Surface hardness 2: how easily the surface is plowed by a hard particle.
-
Micro-crack resistance: how easily a small groove turns into a crack.
-
Stress corrosion resistance: how fast tiny cracks grow in humid air and wash water.
-
Surface durability: how stable the outer nanometers stay after water contact.
Where alumina sits in the control stack
| Control lever | What it changes | Typical impact on scratches | What buyers should ask for |
|---|---|---|---|
| Al₂O₃ in glass composition | Network strength and surface stability | Medium improvement, more stable baseline | Target range + property proof |
| Hot-end coating (e.g., tin oxide) | Initial surface protection | Big improvement | Coating type and coverage control |
| Cold-end coating (wax/PE) | Slip and scuff reduction | Big improvement | Coating weight control |
| Annealing quality | Residual stress level | Strong effect on crack growth | Stress measurement targets |
| Line handling and lubrication | Contact severity | Often the biggest factor | Line audit and scuff rate KPIs |
This is why the best technical conversation starts with performance. Then the recipe is tuned to support it.
If you want the cleanest way to explain alumina to a buyer, it is this: Al₂O₃ improves the glass network, and a stronger network gives the surface more resistance to damage and slow crack growth. Now let’s go deeper.
A bottle that survives the line does not rely on one factor. It relies on a system that prevents surface damage and keeps small flaws from turning into failures.
What does Al₂O₃ change in surface microstructure?
A bottle surface can look smooth and still be weak at the microscopic level. Water, alkali ions, and micro-flaws can turn a clean surface into a fragile one.
Al₂O₃ acts as a network former/intermediate that increases structural connectivity, reduces alkali mobility, and helps the surface stay chemically stable. This often raises hardness and reduces water-driven weakening, which lowers the chance that a scratch becomes a growing crack.
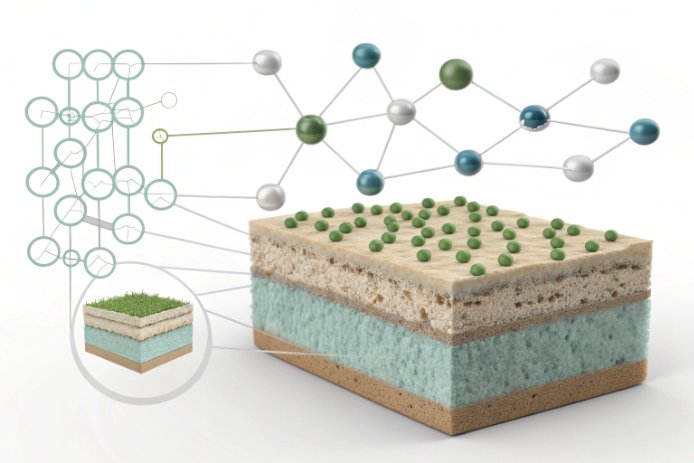
Glass is amorphous, so it does not have grains like metal. Still, it has short-range structure. In soda-lime container glass 3, silica tetrahedra 4 form the backbone, and modifiers like Na₂O and CaO change how open that network is. Alumina enters and changes the balance. It often forms AlO₄ units that connect into the network. This typically reduces the number of “easy pathways” for ions and water to move.
That matters because the bottle surface is not static. In humid air and in rinse water, the surface can develop a thin altered layer. Alkali can leach out. Water can diffuse in. This can lower surface strength and make crack tips more active. When alumina is higher, the network is usually tighter, so alkali release can slow down and the altered layer can be less damaging. In simple terms, the surface stays stronger for longer.
Al₂O₃ can also influence how the surface responds to friction. A tighter network can raise hardness and elastic modulus in a way that reduces the depth of plowing under the same contact load. A scratch that is shallower is less likely to turn into a critical flaw.
Still, alumina is not free. If alumina rises too much in container glass, viscosity rises and the melting window shifts. So the microstructure benefit must be balanced against melting and forming reality.
How alumina changes key surface behaviors
| Surface behavior | What happens when Al₂O₃ increases (within a practical range) | Why it matters for scratches |
|---|---|---|
| Ion mobility | Alkali ions move less easily | Less surface weakening over time |
| Hydration sensitivity | Surface reacts more slowly with water | Slower crack growth in wet conditions |
| Hardness response | Surface resists plowing better | Shallower scratches and fewer deep grooves |
| Crack-tip stability | Stress corrosion slows | Scratches are less likely to “run” into breaks |
A buyer does not need to memorize glass chemistry. A buyer needs a supplier who can show that the surface stays stable under real humidity and line conditions.
Why does alumina raise abrasion resistance on filling lines?
Abrasion on a filling line is not a lab scratch test. It is repeated contact with glass, metal guides, plastic rails, and sometimes hard particles from dust or label debris.
Alumina raises abrasion resistance because it improves baseline hardness and reduces the rate of moisture-assisted crack growth. On filling lines, this helps bottles tolerate repeated rub and contact without turning small scuffs into strength-killing flaws.

Filling lines create a special type of damage. Bottles do not get one scratch. They get thousands of tiny touches: bottle-to-bottle, bottle-to-rail, bottle-to-starwheel. Each touch can create a micro-scuff. Many micro-scuffs together create haze and dullness. Some scuffs become deeper scratches. The deeper ones become strength defects.
Alumina helps in two ways that show up in real production.
First, it improves the baseline mechanical response. When a hard contact point slides on glass, it can either glide or dig. A network that resists deformation reduces digging. That reduces the depth and severity of damage for the same contact force.
Second, it improves the time-dependent part of damage. Many bottle cracks grow by stress corrosion 5, which is driven by water at the crack tip. Filling lines have water, humidity, and sometimes caustic rinses. A surface that is more chemically stable slows this crack growth. That means a scratch is less likely to turn into a delayed break during packing, palletizing, or transport.
But alumina’s advantage can be erased by poor line conditions. If the line has hard contamination particles, even strong glass will scratch. If cold-end coating 6 is inconsistent, the glass will scuff. If rails are worn or misaligned, damage will rise fast.
So, the correct way to use alumina is to set it as part of a package:
-
stable Al₂O₃ range in the glass,
-
stable hot-end and cold-end coating weights,
-
controlled line lubrication,
-
and a scuff-rate KPI at the cold end.
Where the “alumina effect” is strongest on the line
| Line zone | Typical damage mechanism | How alumina helps | What else must be controlled |
|---|---|---|---|
| Transfer points | Impact + sliding | Reduces crack growth from small flaws | Alignment and cushioning |
| Accumulation tables | Bottle-to-bottle rub | Higher baseline abrasion tolerance | Lubrication and bottle spacing |
| Guides and rails | Continuous sliding | Shallower scratches at equal load | Rail material and wear |
| Packing | Scuffs + compression | Slower flaw growth | Case packer settings and separators |
When buyers ask for “scratch resistance,” the best answer is not a single chemistry claim. It is a tested line-performance target supported by composition and coatings.
How to target alumina percentages without hurting formability?
It is easy to say “add more Al₂O₃.” It is harder to melt it cleanly, form it consistently, and keep defects low at scale.
Most soda-lime container glasses use Al₂O₃ in a narrow practical band (often around ~1–3 wt% in many recipes). The safest way to target alumina is to set a property goal, then adjust Al₂O₃ with compensating changes in alkali and alkaline earths while monitoring viscosity, liquidus, and devitrification risk.

In container production, formability means the glass must behave predictably from furnace to feeder to mold. Alumina affects that because it influences viscosity 7 and the temperature-viscosity curve. A higher alumina level often increases viscosity at a given temperature. That can demand higher furnace temperature or different forming temperature. Both can increase energy, wear, and defect risk if not managed.
Alumina can also increase the risk of devitrification 8 in some composition windows. If the liquidus temperature rises too close to the working temperature, crystals can form in the forehearth or during forming. That can show up as stones, cords, or haze. A buyer does not see “too much alumina.” A buyer sees “more defects” and “more breakage.”
The right method is to control alumina with a balanced recipe approach:
-
Increase Al₂O₃ only as far as the furnace and forehearth can support.
-
Use stable raw material sources, because alumina often comes from feldspar 9, nepheline syenite, or alumina-bearing materials that can also carry iron or other impurities.
-
Adjust Na₂O/K₂O and CaO/MgO to keep the melt workable and the chemical durability stable.
-
Verify viscosity points and liquidus in trials, not only in spreadsheets.
For procurement, the cleanest spec is not “Al₂O₃ must be 2.8%.” The cleanest spec is:
-
required scuff performance,
-
required defect limits,
-
required color and durability,
-
then a composition window the supplier can repeat.
A control table for recipe targeting
| Recipe knob | If it increases | Benefit | Risk | How to control the risk |
|---|---|---|---|---|
| Al₂O₃ | Viscosity and network strength | Better durability and abrasion baseline | Harder melting, devitrification | Check liquidus and viscosity curve |
| Na₂O/K₂O | Melting ease | Lower melt temperature | Higher expansion, lower durability | Balance with Al₂O₃ and CaO/MgO |
| CaO/MgO | Stability and stiffness | Better durability and working stability | Liquidus shifts | Keep balanced ratios |
| Cullet % | Faster melting | Lower energy and emissions | Composition variability | Tight cullet sorting and chemistry trend |
A supplier who can show viscosity control, forehearth stability, and defect rates is the supplier who can use alumina safely.
Are nano-additives enhancing scratch resistance in glass?
Many buyers hear “nano” and expect a miracle. In bottles, the most useful nano approach is usually not mixing nanoparticles into the melt. It is adding an ultra-thin surface layer or advanced coating.
Nano-scale coatings and surface treatments can enhance scratch resistance by reducing friction and protecting the outer surface. Bulk nano-additives inside the melt are less common in bottle glass because they can raise cost and defect risk, but thin nano-coatings (sol-gel, plasma, ALD-like films) are gaining attention for premium and returnable applications.

There are two different “nano” stories in glass bottles.
1) Nano inside the glass (bulk additives)
Putting nanoparticles into a large furnace melt sounds attractive, but it is difficult in container production. The reasons are simple:
-
Dispersion is hard at industrial scale.
-
Agglomerates can become stones or seeds.
-
Many nano oxides are expensive.
-
Some additives change color, redox, or refining behavior.
For high-volume bottles, bulk nano-additives usually struggle to beat the cost and stability of classic composition tuning plus coatings.
2) Nano on the surface (thin protective layers)
Surface treatments are more realistic because they target the part that scratches: the outer microns. Thin layers can reduce friction and make the surface less vulnerable to abrasion. Some premium systems use:
-
alumina or mixed oxide nano-layers,
-
plasma-enhanced coatings,
-
very thin barrier films on returnable bottles.
These approaches can reduce scuffing and improve “gloss retention” after repeated handling. They also can help in reuse loops where bottles are washed and returned many times. That is where scratch resistance becomes a lifetime cost driver.
But coatings bring their own controls:
-
coating adhesion and uniformity,
-
compatibility with labels and inks,
-
food-contact safety for any surface that can contact product,
-
durability through washing and caustic cycles.
So the procurement rule is: if a supplier offers a nano coating, ask for performance data that matches your line and your washing conditions, and ask for the compliance statement for the destination market.
A simple decision table for nano options
| Nano approach | Best use case | Benefit | Main risk | What to request from supplier |
|---|---|---|---|---|
| Bulk nano-additives in melt | niche specialty glass | Potential hardness and durability gains | defects and cost | defect data + long-run stability proof |
| Sol-gel nano coatings | premium cosmetics, reuse | lower scuffing, better gloss | coating wear and adhesion | abrasion cycles + adhesion tests |
| Plasma/ALD-like films | high-end or returnable | strong thin protection | capex and process complexity | lifetime testing + wash resistance |
| “Nano” marketing only | none | none | wasted budget | measurable test results or avoid |
Scratch resistance in glass bottles improves most when surface protection is real, measurable, and repeatable. Nano coatings can help, but only when they come with a serious control plan.
Conclusion
Al₂O₃ supports scratch resistance by strengthening the glass network and stabilizing the surface. The best results come from balanced composition, strong coatings, and clean line handling.
Footnotes
-
Scratch resistance: A material property describing the ability to withstand surface abrasion and deformation without failing. ↩
-
Surface hardness: The resistance of a material’s surface to permanent indentation or plowing by external objects. ↩
-
Soda-lime container glass: The most common glass type for packaging, offering a balance of cost, clarity, and chemical stability. ↩
-
Silica tetrahedra: The fundamental SiO₄ structural units that form the backbone of the glass network. ↩
-
Stress corrosion: A failure mechanism where cracks grow due to the combined effects of tensile stress and a corrosive environment. ↩
-
Cold-end coating: A lubricious layer applied after annealing to reduce friction and protect bottles from scuffing. ↩
-
Viscosity: A measure of a fluid’s resistance to flow, which dictates how glass behaves during melting and forming. ↩
-
Devitrification: The uncontrolled crystallization of molten glass during cooling, which can cause structural defects. ↩
-
Feldspar: A common aluminosilicate mineral used as a raw material source for alumina in glass production. ↩
-
Sol-gel: A chemical process used to produce high-performance surface coatings from liquid precursors. ↩








