Weak bottles do not fail in the lab. They fail on conveyors, in cases, and at customer sites, where one crack becomes a full recall.
Yes, small ZrO₂ additions can raise hardness and sometimes improve real bottle strength, but only when the melt stays clean and fully dissolved—otherwise ZrO₂ becomes stones, cords, and higher rejects.

A practical way to think about ZrO₂ in container glass?
ZrO₂ can help, but it is not a simple “add strength” button. A glass bottle breaks because surface flaws grow into cracks. So the strongest bottle is often the one with the smoothest surface and the lowest defect count, not the one with the highest lab modulus number. ZrO₂ can still play a role because it is a stiff, high-field-strength oxide. It can tighten the glass network, increase hardness, and reduce crack growth in some compositions. That can lift the average performance in scratch and impact tests.
But ZrO₂ is also famous for one hard truth: it is easy to turn into stones if it does not dissolve. In soda-lime container furnaces, any undissolved zircon (ZrSiO₄) 1 grains, refractory spalls, or zircon-rich carryover can become visible inclusions. Those inclusions act like pre-made crack starters. So ZrO₂ can improve “intrinsic” strength while hurting “real” strength if the plant cannot melt and refine it cleanly.
When I evaluate ZrO₂ for bottles, I start with three questions:
1) Is the current breakage driven by glass chemistry or by defects and handling?
2) Can the furnace and forehearth hold enough temperature and mixing to fully dissolve Zr-bearing additions?
3) Can cullet and raw materials be kept clean enough so ZrO₂ does not come with extra stones?
If the main issue is defects, I fix fining, temperature stability, and cullet first. If the melt is already clean and stable, then ZrO₂ becomes a real tuning tool.
| Decision area | What ZrO₂ can improve | What ZrO₂ can worsen | What to control first |
|---|---|---|---|
| Intrinsic properties | hardness, elastic modulus, crack resistance | viscosity drift if pushed too far | base SiO₂–Na₂O–CaO–MgO stability |
| Real bottle strength | fewer scratches can mean fewer failures | stones and cords can raise rejects | fining stability, forehearth cold spots |
| Appearance | sometimes better brilliance if defects drop | haze and seeds if dissolution fails | raw material size distribution, mixing |
| Cost and risk | longer bottle life in returnables | higher batch cost and supply risk | proof of yield improvement |
This framework keeps the project grounded. It also prevents a common mistake: adding ZrO₂ to solve breakage that is really caused by poor annealing 2, bad handling, or dirty cullet.
Now, the real question is dosage. A lot of teams ask for a number, but the “right” number depends on what the furnace can dissolve.
A bottle recipe is only as good as the plant can run it. So I move from theory to production windows next.
What ZrO₂ dosage (e.g., ~0.2–1.0 wt%) actually boosts modulus of rupture and impact strength in soda-lime glass?
When dosage is too low, nothing changes. When dosage is too high, melting slows and stones appear. That is why most ZrO₂ programs fail.
In many soda-lime bottle bases, ~0.2–0.6 wt% ZrO₂ is the most practical “benefit window,” while ~0.6–1.0 wt% can work only if melting and refining are strong enough to keep Zr fully dissolved and defect-free.
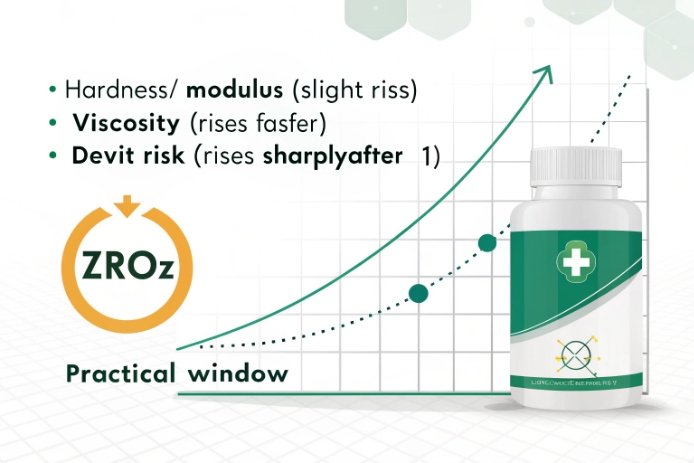
ZrO₂ works like a stiffener. At small levels it can raise hardness and sometimes improve modulus of rupture (MOR) 3 in lab bars. But bottle impact strength is not only MOR. It is MOR plus surface quality, plus stress state, plus the flaw population from forming. So the “dose that helps” is the dose that improves intrinsic behavior without creating new flaws.
A practical trial path usually looks like this:
- Start at 0.2 wt% ZrO₂ (or equivalent Zr contribution) replacing part of the silica or alumina source on a molar plan, not just adding extra solids.
- Run long enough to reach steady-state in the furnace and cullet loop.
- Measure seeds, stones, cords, and surface scratch response, not only MOR.
In my experience, the benefit curve often looks like “fast early gain, then flatten.” That means:
- 0.2 → 0.4 wt% can show a noticeable change in hardness and surface durability 4.
- 0.6 wt% can still help if the melt stays clean.
- 0.8–1.0 wt% is where stone risk rises sharply in many container furnaces, unless the plant is designed for it.
The biggest trap is to judge success after a short run. ZrO₂ returns in cullet. If cullet rises over time, Zr can creep upward. A recipe that starts at 0.6% can quietly become 0.75% after weeks if the mass balance is not tracked.
| ZrO₂ level (wt%) | Likely outcome | Main risk | Best KPI to judge it |
|---|---|---|---|
| 0.0–0.2 | minimal intrinsic change | false “no effect” conclusion | hardness trend, scratch trend |
| 0.2–0.6 | best balance for many plants | small viscosity shift | seed count + reject rate + scuff score |
| 0.6–1.0 | possible extra gain if clean melt | stones, cords, higher liquidus | stone/cord incidents + finish defects |
| >1.0 | rarely worth it in standard bottles | high defect risk and cost | total cost per good bottle |
If the goal is impact strength in real distribution, I treat defect rate as the primary metric. If ZrO₂ improves MOR but raises stones by 20%, the real bottle strength program loses.
Next comes the comparison question. Many plants can get 70% of the strength benefit from Al₂O₃ with much less risk.
How does ZrO₂ compare with Al₂O₃ or ZnO in raising hardness and elastic modulus without hurting formability?
Teams love ZrO₂ because it sounds “premium.” But premium does not matter if the line slows down.
ZrO₂ can raise hardness and stiffness efficiently per unit addition, but Al₂O₃ is usually the safer, cheaper strength-and-durability lever for bottle plants, while ZnO is a niche modifier that can help durability and surface behavior but needs tight dosing control.
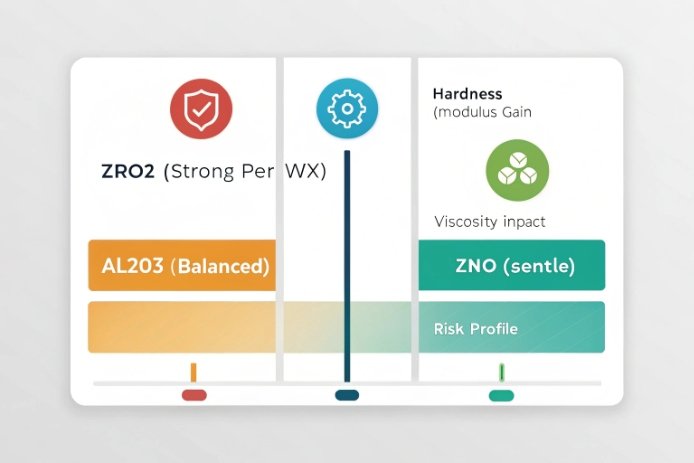
Al₂O₃ is the workhorse intermediate oxide in container glass. It strengthens the network, improves chemical durability, and often increases hardness with fewer melting surprises, especially when increased within a modest range. Many bottle plants already use alumina-bearing raw materials, so raising Al₂O₃ by 0.3–0.8 wt% can be straightforward.
ZrO₂ is more powerful per unit, but it is less forgiving. It can increase viscosity and can create undissolved particles if raw material grain size or mixing is weak. So the “formability” risk is higher. If the working range 5 shifts, gob temperature targets move, and finish tolerances can drift.
ZnO is different. It acts more like a modifier with some intermediate behavior. It can improve durability and can sometimes help reduce certain surface corrosion patterns. In some compositions it can also help reduce surface crystallization tendencies. Still, ZnO is rarely used as a main strength lever in high-volume bottles because of cost and the need for very stable dosing.
A clean way to compare is to evaluate three outputs:
- change in viscosity curve (forming stability)
- change in defect population (seeds/stones/cords)
- change in surface hardness and scuffing (real handling)
| Additive | Strength/hardness effect | Formability risk | Stone/devit risk | When it is the best choice |
|---|---|---|---|---|
| Al₂O₃ | steady improvement, good durability | low to medium | low | need safer durability + strength, low risk |
| ZrO₂ | strong stiffness/hardness potential | medium to high | medium to high | clean furnace, tight QC, premium bottle targets |
| ZnO | moderate durability and surface benefits | medium | low to medium | specific corrosion/scuff needs, controlled cost |
| B₂O₃ (context) | helps meltability and can reduce devit | medium | low to medium | when melting/refining is the main limit |
If the plant is lightweighting aggressively, Al₂O₃ is often the first move because it helps both durability and stiffness without creating a new “stones” problem. If the plant already has excellent melting and wants a premium strength boost, ZrO₂ can be worth a trial.
But this choice must also consider devitrification and liquidus, which is where ZrO₂ can become painful.
Does adding ZrO₂ increase liquidus or devitrification risks (zircon/baddeleyite) and lead to stones or cords in production?
A strength additive that causes devit lines is not a strength additive. It becomes a reject generator.
Yes, ZrO₂ can increase liquidus sensitivity and can promote zircon- or baddeleyite-related crystalline issues if local chemistry or cold zones exist, and undissolved zircon particles are a common stone source in production.
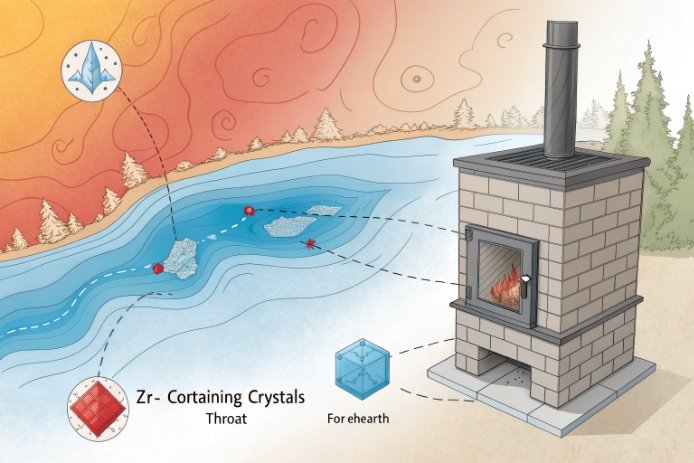
There are two main “Zr problems” in bottle furnaces.
Problem 1: undissolved Zr-bearing raw material.
If Zr enters as zircon sand or a zircon-containing batch component, grain size and residence time matter. Coarse grains can survive as stones. Once a stone exists, it can travel into the bottle wall or sit near the surface. Under impact, it behaves like a crack starter. This is not theoretical. It is one of the most common reasons Zr trials fail.
Problem 2: crystallization in cold or stagnant zones.
Even when Zr is dissolved, it can change the liquidus behavior and can raise the chance of crystals forming on cold surfaces. In soda-lime systems, Zr can participate in zircon-type phases when silica is available, and baddeleyite (ZrO₂) 6 can appear when conditions favor it. In normal container glass, these phases do not usually grow fast in the bulk melt, but they can grow on surfaces where temperature is lower and residence time is high.
This is why forehearth profile matters more than people expect. A small cold corner can turn a “safe” recipe into a devit recipe. Also, cords 7 can appear when local chemistry bands exist. If Zr distribution becomes uneven (poor mixing, batch carryover, redox/foam issues), cords and striae can increase. Then the bottle looks worse and breaks sooner.
| Risk trigger | What it creates | Why it happens | Fix that works in real plants |
|---|---|---|---|
| coarse zircon grains | stones | incomplete dissolution | finer raw material, longer melt time, better mixing |
| forehearth cold spots | devit skins | low wall temp + long residence | eliminate dead zones, stabilize profile |
| unstable fining/foam | cords | chemistry bands and poor homogenization | steady SO₃ feed, stable pull, stable combustion |
| refractory wear (AZS/zircon parts) | Zr-rich inclusions | spalls fall into melt | wear monitoring, block replacement timing |
| high Zr + high local Si | zircon-like crystals | local phase stability shifts | cap Zr level, improve homogeneity |
If ZrO₂ is used, the control plan must include stone monitoring and a defined “stop rule.” A plant should not wait for customer claims. It should stop when stone rate or devit line count crosses an internal limit.
Now the final question is about trade-offs. Even if ZrO₂ works technically, it must also win on cost and process risk.
What are the melt/refining, refractory-wear, and cost trade-offs of ZrO₂—and when is Al₂O₃/B₂O₃ a better choice?
A recipe that makes one bottle stronger but makes the furnace harder to run is not a real win. The win is cost per good bottle.
ZrO₂ can raise stiffness and hardness, but it increases raw material cost and can raise stones and refractory-wear sensitivity. Al₂O₃ is often the safer strength tool, and B₂O₃ is often better when the real bottleneck is meltability and refining rather than intrinsic strength.

Melt and refining trade-offs
ZrO₂ can raise viscosity and can require more energy or a different temperature profile to reach the same fining 8 level. If the furnace already runs near capacity, this can reduce pull or raise fuel use. Also, if Zr increases seed retention or changes foam behavior in a specific plant, yield can drop. ZrO₂ is most successful when the furnace has extra thermal headroom and strong mixing.
Refractory and wear trade-offs
Zr in the system often links to refractory issues because zircon and zirconia are used in many refractories 9. If wear increases, Zr-rich spalls can enter the melt and appear as stones. Also, higher alkali and sulfate environments already stress crowns and superstructures. If a ZrO₂ trial forces higher operating temperatures, refractory wear can accelerate.
Cost and supply trade-offs
Zr-bearing raw materials and high-purity zirconia can be expensive compared with alumina sources. For a beverage or standard food bottle, the cost case is hard unless reject reduction is large. For premium spirits or perfume-style heavy bottles, the value case can be stronger, because customer tolerance for “tiny stones” is near zero and the price per bottle is higher.
When Al₂O₃/B₂O₃ is a better choice
- Choose Al₂O₃ when the goal is better durability and a safer stiffness upgrade with low stone risk.
- Choose B₂O₃ when the goal is better meltability, lower liquidus, and fewer defects from incomplete melting or devit, especially in hot-fill or returnable systems where surface quality is the real strength driver.
| Goal | Best first lever | Second lever | When ZrO₂ earns a place |
|---|---|---|---|
| Lightweighting with stable forming | Al₂O₃ | MgO/CaO balance | only after defect rate is already low |
| Reduce breakage from scratches/scuff | hot-end/cold-end control + Al₂O₃ | ZnO (small) | when hardness gains reduce scuff in handling |
| Reduce stones/devit rejects | B₂O₃ (small) + process fixes | Al₂O₃ | rarely; ZrO₂ can worsen this |
| Premium strength and feel | Al₂O₃ + tight QC | ZrO₂ (small) | when furnace can dissolve it cleanly |
| Improve chemical durability | Al₂O₃ | ZrO₂ (small) | when extractables need extra margin |
A simple rule keeps projects safe: if the plant cannot guarantee low stones and stable forehearth temperature, ZrO₂ is not the right first move. In that case, Al₂O₃ and B₂O₃ deliver more reliable gains with less production risk.
Conclusion
ZrO₂ can improve bottle strength at small doses, but it only works when melting and homogeneity are excellent; otherwise Al₂O₃ or B₂O₃ delivers safer, cheaper performance gains.
Footnotes
-
A mineral form of zirconium silicate, often used as a raw material or refractory. ↩
-
Heat treatment to remove internal stresses; critical for preventing bottle breakage. ↩
-
A measure of a material’s strength before it breaks under bending load. ↩
-
Resistance of the glass surface to chemical attack and physical wear. ↩
-
The viscosity range where glass can be formed into bottles without defects. ↩
-
A natural mineral form of zirconium dioxide, sometimes appearing as defects. ↩
-
Visible lines of inhomogeneity in glass that weaken the bottle structure. ↩
-
The process of removing gas bubbles from molten glass to ensure clarity. ↩
-
Heat-resistant materials used to line glass furnaces, often containing zirconia. ↩








