Uneven frosting, stains, and weak bottles can turn a premium “matte” project into a return nightmare. The cause is often not the etch tank.
Glass chemistry controls how fast acids attack the surface, how uniform the frost looks, and how much strength is lost, so the right composition makes etching predictable and repeatable.

What acid etching really “sees” on a bottle surface
Acid etching 1 is not just a surface decoration step. It is a controlled corrosion step. Most commercial frosting for bottles relies on fluoride chemistry (often hydrofluoric acid 2 based systems or fluoride donors), because standard mineral acids do not dissolve silica fast enough. The key reaction removes parts of the silica network and leaves a micro-rough surface that scatters light. That micro-roughness is what the eye reads as “frost density.”
Composition decides how easy it is for the acid to break the network and how fast ions leave the surface. A soda-lime glass 3 bottle is a silica network held together and “opened up” by modifiers. More modifiers (Na₂O, K₂O, CaO, MgO) generally make the network easier to attack. More network formers or intermediates (SiO₂, Al₂O₃) generally make it harder to attack. That is why two bottles can sit in the same etch bath for the same time and still come out with different haze, different feel, and different stain risk.
Why etch rate and frosting density are not the same thing
A fast etch rate can still produce a weak-looking frost if the surface dissolves smoothly. A slower etch can create a strong frost if it creates a uniform micro-pit pattern. So I separate etch performance into three outputs:
-
Etch rate (material removed per minute)
-
Frost density (haze and gloss drop)
-
Surface roughness (Ra, Rz, and feel)
Where defects come from
Etch defects usually come from non-uniform surface chemistry:
-
Hot-end coatings can block etch and create “holidays”
-
Cords and stones create bands or pinholes
-
Salts and residues create stains and crater-like marks
-
Redox drift changes colorants and surface deposits, which changes how the acid wets
The simplest way to manage it
A stable etch program matches three things:
1) A glass recipe that etches at a controlled speed
2) A furnace and lehr process that keeps the surface consistent
3) Etch line controls (bath chemistry, temperature, rinse, and drying) that prevent stains and micro-cracks
| What you want | Chemistry direction | Why it helps | What can go wrong |
|---|---|---|---|
| Faster etch | More alkali/modifiers | Network opens easier | More stain risk, more strength loss |
| Slower, controlled etch | Higher SiO₂ + Al₂O₃ | Higher durability | Longer cycle time, uneven frost if hot-end blocks |
| Cleaner frost | Stable melt, low cords/devit | Uniform surface response | Requires strong cullet and refining control |
| Better post-etch adhesion | Low salts, stable alkalinity | Better wetting and coupling | Needs rinse/dry control and surface energy checks |
If the goal is a premium, repeatable frosted glass 4 at scale, I always start with oxide balance and surface consistency, not with “stronger acid.”
Now I will break it down by oxide levels, colorants, furnace and hot-end factors, and strength targets after etch.
The details below are written to help batch and decoration teams speak the same language.
Which oxide levels (SiO₂–Na₂O–CaO–MgO–Al₂O₃, B₂O₃) most control etch rate, frosting density, and surface roughness?
When etching drifts, the fastest clue is usually oxide balance. The surface reacts like a chemical map of the glass network.
Higher SiO₂ and Al₂O₃ usually slow etch and tighten roughness, while higher Na₂O/K₂O and total modifiers usually speed etch and raise roughness and haze—so the best frosting comes from a controlled middle, not extremes.
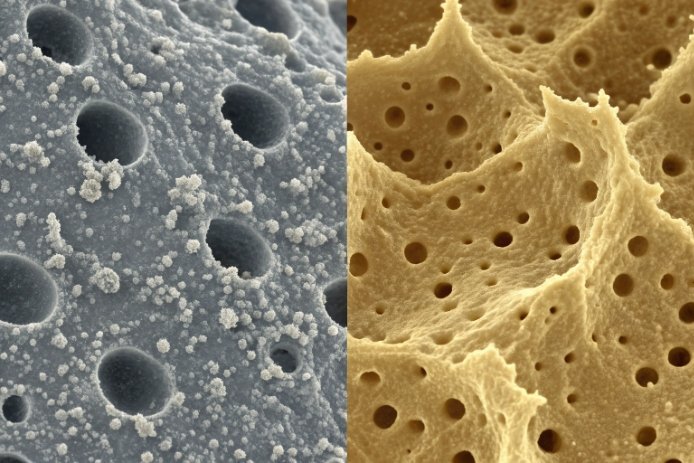
SiO₂: the main “brake”
Silicon dioxide 5 (SiO₂) builds the network. More SiO₂ usually lowers etch rate and makes frosting take longer. It can also produce a more “silky” frost if the bath is controlled, because the surface dissolves more evenly. If SiO₂ rises too much, the etch time can become long, and small surface differences (like hot-end coatings or cords) become more visible as non-uniform frost.
Na₂O (and K₂O): the main “accelerator”
Sodium oxide 6 (Na₂O) breaks up the network. More Na₂O usually raises etch rate and makes frost develop faster. It can also increase surface alkalinity after rinsing if ions remain, which raises stain risk. In my experience, an aggressive alkali level can make the frost look strong, but it can also make it harder to control batch-to-batch, especially when cullet quality changes.
CaO and MgO: stability levers that also shape the etch texture
CaO and MgO are modifiers, but they often change the texture more than the speed. A balanced CaO/MgO can reduce “muddy” frosting and help uniform micro-pitting. Too much MgO in some container recipes can raise crystallization risk, and crystals can etch as pinholes.
Al₂O₃: durability and uniformity
Aluminium oxide 7 (Al₂O₃) often improves chemical durability and reduces ion exchange. For etching, that usually means slower etch, but better repeatability. A controlled Al₂O₃ level can make the surface less sensitive to humidity storage before etch, which reduces stain surprises.
B₂O₃: can change the window
B₂O₃ can increase chemical complexity and can shift etch response. In some designs it helps make frosting develop faster at a given bath condition, but it can also change haze tone and require a tighter rinse and neutralization plan.
| Oxide lever | Etch rate trend | Frost density trend | Roughness (Ra) trend | Practical note |
|---|---|---|---|---|
| SiO₂ ↑ | Down | Can stay high if time increases | Often smoother | Long cycle, highlights surface differences |
| Na₂O/K₂O ↑ | Up | Up fast | Rougher | Higher stain risk if rinse is weak |
| CaO ↑ | Mixed | Mixed | Mixed | Often stabilizes, but watch haze tone |
| MgO ↑ | Mixed | Mixed | Can create sharper micro-pits | Watch devit and pinholes |
| Al₂O₃ ↑ | Down | More controllable | Often tighter | Helps durability and post-etch stability |
| B₂O₃ ↑ | Mixed | Mixed | Mixed | Needs line trials, can shift hue and clarity |
When I set recipe direction for frosting, I aim for a controlled etch speed that gives time for uniform wetting, then I tune bath agitation and temperature to lock in texture.
Do colorants and additives (Fe₂O₃/FeO, Cr₂O₃, TiO₂, ZrO₂) change reaction kinetics, hue shift, and post-etch clarity?
Color glass makes etching harder because the eye judges it more strictly. Small changes in haze and tone show up fast under store lighting.
Colorants usually change etch performance indirectly by changing heat history, surface deposits, and how light scatters after frosting, while TiO₂/ZrO₂ traces can change clarity and pinhole risk if they drift or form inclusions.

Iron redox: Fe²⁺ vs Fe³⁺ changes the look after etch
Iron exists in different states. That shifts green/amber tone. After etch, the surface scatters more light. That can make the bottle look lighter and more “milky.” If redox 8 drifts, the same etch depth can show different perceived color because scattering and absorption interact. In practice, the bath did not change, but the customer sees a color shift.
Cr₂O₃: stable green, but frosting can look darker
Chromium green is strong. After etch, the surface scattering can reduce gloss and increase perceived depth. If the frost is heavy, the bottle can look darker than expected, especially in thick zones. So the recipe and etch target must be set using the same wall thickness the customer will see in real bottles.
TiO₂: opacity and scatter risks if it drifts
Trace TiO₂ can increase light scattering. Under a frosted surface, that can push the look from “matte” to “chalky.” If TiO₂ comes in as undissolved specks from cullet or raw materials, those specks can etch differently and become pinholes.
ZrO₂: durability help, but inclusions are the danger
ZrO₂ can improve durability and reduce ion exchange. That can help post-etch stability and reduce stain risk. But if ZrO₂ arrives as particles or is linked to ceramic contamination, it can act like a stone. Stones often show up as tiny bright dots after etch.
| Additive/colorant | Kinetics effect | Appearance effect after etch | Main risk to control |
|---|---|---|---|
| Fe redox drift | Small direct effect | Hue and brightness drift | Furnace redox stability |
| Cr₂O₃ | Small direct effect | Darker matte look at same haze | Thickness-based color spec |
| TiO₂ trace | Indirect | “Chalky” haze, pinholes if specks | Cullet and raw material purity |
| ZrO₂ trace | Indirect | More stable surface, but stones if inclusions | Contamination control |
My rule is simple: if the project needs tight color, I lock redox and cullet streams first, then I tune etch depth. Doing it in the other order wastes time.
How do cullet quality, furnace redox, and hot-end treatments (SnO₂/SO₂) influence pinholes, stain risk, and coating/printing adhesion after etch?
A perfect oxide recipe still fails if the surface carries residues or hidden defects. Etching exposes everything.
Cullet quality and redox stability decide how clean and uniform the glass surface is, and hot-end SnO₂ or sulfate-related residues can block etch, raise stain risk, and reduce post-etch coating or printing adhesion if the pretreatment is not matched.

Cullet quality: the fastest path to pinholes and “dirty frost”
Dirty cullet 9 can bring ceramics, metals, organics, and mixed-color streams. Ceramics become stones. Stones etch differently and show as pinholes. Organics can shift redox and increase deposits that later become stains. Mixed-color cullet can change iron redox behavior and hue after etch.
Furnace redox: controls deposits and color stability
Redox drift can change:
-
Iron state and color perception after frost
-
Sulfate behavior in refining, which can change surface residues
-
Volatile deposits that land on bottles and cause wetting breaks
A wetting break often turns into a stain after etch because the acid attacks unevenly and rinses unevenly.
Hot-end SnO₂: can create etch “holidays” if not managed
Hot-end coatings improve scratch resistance, but they can act like a shield in etch tanks. If the hot-end layer is thick or uneven, etch rate changes by zone. The result is patchy frosting. That patchiness can also hurt post-etch adhesion for coatings or printing, because surface energy varies.
SO₂ and sulfate carryover: stain and crater risk
Even small ionic residues can cause stains after etch because fluoride systems are sensitive to local chemistry. If rinse water is not strong enough, salts can re-deposit and leave a “shadow” stain after drying.
| Upstream factor | Etch defect it drives | Post-etch risk | Best control action |
|---|---|---|---|
| Ceramic contamination in cullet | Pinholes, bright dots | Peel in coatings, print skips | Sorting + stone count KPI |
| Organics in cullet | Random stains | Adhesion drift | Cullet cleaning + LOI checks |
| Redox swings | Hue drift, stain bands | Batch-to-batch variation | Redox KPI + color spec audit |
| Hot-end SnO₂ uneven | Patchy frost | Uneven wetting and adhesion | Hot-end control + adjusted etch/activation |
When coating or printing follows etch, I always add a “surface reset” step after etch: neutralize, DI rinse, dry, and then activate. That makes results less sensitive to small upstream drift.
What composition targets preserve strength after etching—residual wall thickness, micro-crack suppression, and Ra/adhesion test thresholds?
Etching removes glass. It also changes flaw shape. If the process is too aggressive, the bottle loses strength and fails in top-load or impact.
Strength is preserved when composition supports controlled, uniform etching, when material removal stays inside a defined limit, and when roughness stays high enough for adhesion but low enough to avoid micro-crack growth.

Residual wall thickness: define a maximum removal, not just a time
The safest control is to specify a maximum thickness loss by zone (shoulder, sidewall, heel). Time alone is not reliable because bath activity changes. For lightweight bottles, the margin is small, so the allowable removal must be tighter than for heavy bottles.
Micro-crack suppression: uniform wetting beats “strong acid”
Micro-cracks often come from uneven attack and local stress. The best prevention steps are simple:
-
Keep bath concentration and temperature stable
-
Use consistent agitation to avoid boundary layers
-
Avoid dry spots and trapped bubbles on the surface
-
Rinse and neutralize fast to stop reaction
-
Dry without leaving salts that can pull moisture later
Composition helps because higher SiO₂ and controlled Al₂O₃ reduce uncontrolled attack and reduce ion exchange. But if the recipe gets too durable, teams push the bath harder, and that can create roughness spikes and strength loss. So the target is a controlled etch rate that does not force aggressive conditions.
Roughness targets: enough for adhesion, not so much that strength collapses
After etch, coatings and printing often adhere better because the surface has higher area. But if surface roughness 10 (Ra) is too high, sharp pits can act as crack starters. A stable program sets:
-
A target Ra window by product
-
A haze or gloss target for appearance
-
Adhesion targets for any post-etch decoration
Practical composition directions that protect strength
I use simple direction targets:
-
Keep SiO₂ high enough to avoid runaway attack
-
Keep Al₂O₃ at a durable baseline (often ≥1–2% is a useful practical floor)
-
Keep total alkali stable and not excessive to reduce ion exchange under humidity
-
Keep CaO/MgO balanced to reduce devit and pinhole risk
| Target | Why it matters | Suggested threshold style (set by your line trials) |
|---|---|---|
| Max thickness loss (µm) | Protects burst/top-load | Limit by zone, not only average |
| Ra window (µm) | Balances adhesion vs strength | Define min for adhesion, max for strength |
| Tape/cross-hatch after humidity | Catches delayed failures | Test after 24–72h humidity aging |
| Abrasion/rub cycles | Confirms durability of frosted surface and coatings | Dry and wet rub limits |
| Pinhole count / stain rating | Protects mirror look and print yield | Visual spec per dm² and zone |
If a customer wants a very heavy frost on a lightweight bottle, I prefer to change the design (more wall in critical zones) or use a two-step finish (milder etch plus coating) rather than pushing the acid until strength drops.
Conclusion
Composition controls etch speed, frost texture, stains, and strength loss. A stable oxide window, clean cullet, steady redox, and clear Ra/thickness limits make acid-etched bottles repeatable at scale.
Footnotes
-
Glass etching is the technique of creating art on the surface of glass by applying acidic, caustic, or abrasive substances. ↩
-
Hydrofluoric acid is a solution of hydrogen fluoride in water, a precursor to almost all fluorine compounds, and is used to etch glass. ↩
-
Soda-lime glass is the most prevalent type of glass, used for windowpanes and glass containers for beverages and food. ↩
-
Frosted glass is produced by the sandblasting or acid etching of clear sheet glass. ↩
-
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula SiO₂, most commonly found in nature as quartz. ↩
-
Sodium oxide is a chemical compound with the formula Na₂O, used in ceramics and glasses. ↩
-
Aluminium oxide is a chemical compound of aluminium and oxygen with the chemical formula Al₂O₃. ↩
-
Redox is a type of chemical reaction in which the oxidation states of atoms are changed. ↩
-
Cullet is recycled glass that is crushed and ready to be remelted. ↩
-
Surface roughness is a component of surface texture, quantified by the deviations in the direction of the normal vector of a real surface from its ideal form. ↩








