Green bottles feel “easy” until the shade shifts mid-run. Then the whole week turns into sorting, re-melting, and explaining pallets.
Most green bottles use a standard soda-lime base (SiO₂–Na₂O–CaO–MgO) and then tune green by controlling iron valence (Fe²⁺/Fe³⁺) plus a small Cr₂O₃ add. The real secret is stable redox, not just a fixed recipe.

The baseline green bottle recipe and why it stays stable
Start with a “container soda-lime” base, then color on top
In mass production, green bottles rarely reinvent the full chemistry. Most plants lock a container-friendly soda-lime base 1 so forming and durability stay consistent. Then color is added with iron and chromium, and the hue is fine-tuned with redox control 2 (how oxidizing or reducing the melt is). This approach keeps viscosity 3, working range, and annealing 4 behavior stable across SKUs, while the color team adjusts only a small set of levers.
A practical baseline for continuous furnaces is:
-
High SiO₂ for structure and chemical durability
-
Na₂O for melt workability and cost control
-
CaO and MgO for durability and stiffness, plus better forming stability
From there, the “green” is mostly:
-
Iron total (how much tint you have)
-
Iron valence (how much of that tint is blue-green vs yellow-green)
-
Chromium level (how deep and how “emerald” the green looks)
Redox is the main reason two “same recipes” look different
Two batches can have the same Fe₂O₃ and Cr₂O₃ numbers and still look different. That happens when the furnace redox shifts and changes Fe²⁺/Fe³⁺ balance. More Fe²⁺ pushes the glass toward a cooler, deeper green. More Fe³⁺ pushes it warmer and more yellow-green. Chromium also responds to redox and can slide the hue if the melt gets too oxidizing.
Use a simple control mindset: fixed base + small trims
When the base stays fixed, the team can treat colorants like trims and keep changes small. That reduces cords, streaks, and batch-to-batch noise.
| Layer | What stays fixed | What gets tuned | What the customer notices |
|---|---|---|---|
| Base glass | SiO₂–Na₂O–CaO–MgO window | small Al₂O₃/K₂O trims | forming stability, strength |
| Color package | target Fe + Cr bands | Fe²⁺ fraction via redox | green hue and depth |
| Process window | residence + mixing | burner balance + fining | cords, haze, shade drift |
If the buyer wants “consistent green,” the best promise is not a single composition line. It is a process plan that keeps redox and cullet stable.
Now I will break this down into the four questions that usually decide success in production and in purchasing.
Green looks forgiving, but buyers still measure it. So the recipe needs numbers and control points.
What oxide percentages (SiO₂–Na₂O–CaO–MgO) and colorants (Fe₂O₃/FeO, Cr₂O₃) are typically used for standard green?
Green bottles sell well because they hide small raw material swings better than flint. Still, “standard green” needs a clear target band.
A typical standard green bottle uses a soda-lime base around ~70–74% SiO₂, ~12–15% Na₂O, ~8–11% CaO, and ~2–4% MgO, then colors with roughly a few-tenths percent total iron plus a few-hundredths to few-tenths percent Cr₂O₃. The Fe²⁺ share controls whether the green looks cool or yellowish.

Typical base-oxide window used for continuous container production
Most green container lines keep the base close to common soda-lime container chemistry so melting, forming, and annealing remain predictable. In practical terms, the base is a “container recipe,” and green is a color package on top of it.
A production-friendly band that matches many commercial container glasses is:
-
SiO₂: ~70–74 wt%
-
Na₂O: ~12–15 wt%
-
CaO: ~8–11 wt%
-
MgO: ~2–4 wt%
Small supporting oxides (like Al₂O₃ 5 and K₂O) still matter for durability and workability, but they are not the main color drivers.
Typical “standard green” colorant bands
For standard green, iron provides the main absorption 6, while chromium sharpens and deepens the green. A common industrial reference point for commercial green container glass uses chromium oxide and iron oxide additions in the few-tenths percent range. In daily plant control, I focus on:
-
Total Fe (reported as Fe₂O₃): sets overall tint depth
-
Fe²⁺ portion (often expressed as FeO equivalent or Fe²⁺/Fe_total proxy): sets cool vs warm hue
-
Cr₂O₃: strengthens green and pushes toward emerald character
What shifts the hue most in “standard green”
-
Higher Fe²⁺ fraction → cooler, deeper green
-
Higher Fe³⁺ fraction → warmer, more yellow-green
-
Dirty cullet organics → redox swings → cords and shade waves
| Target look | Fe₂O₃ total (typical band) | Fe²⁺ share (direction) | Cr₂O₃ band | Risk to watch |
|---|---|---|---|---|
| Standard green | ~0.2–0.5% | moderate Fe²⁺ | ~0.05–0.30% | drift if redox swings |
| Darker standard green | higher end of Fe | slightly higher Fe²⁺ | higher end of Cr | cords if mixing is weak |
In practice, the best “standard green” recipe is the one that tolerates your cullet reality. That is why emerald and olive are not just different colorant levels. They are different redox strategies.
How do batch ratios differ between emerald green and olive green bottles in mass production?
Emerald and olive can both be “green,” but buyers see them as different products. The furnace also treats them as different control problems.
Emerald green is usually a more chromium-driven, cleaner green that needs a stable (often more oxidized) environment for the desired Cr(III) look, while olive green is often more iron- and reduced-sulfur-driven and relies on a more reducing redox balance to land the warmer tone and stronger light protection.

Emerald green: chromium-forward, cleaner hue
Emerald green in mass production is often built by raising Cr₂O₃ and holding iron and redox steady so the green stays vivid and not muddy. The batch usually does not need “special” base oxides. It needs:
-
a consistent Cr₂O₃ feed,
-
controlled iron baseline (from cullet and sand),
-
stable redox so chromium stays in the desired coloring state.
Many teams also avoid strong reduction because it can create unwanted shade shifts and can worsen cords when gradients appear.
Olive green: iron + reduced sulfur character, warmer tone
Olive green often leans on higher iron and a controlled reduced sulfur contribution. This is why olive can look warmer and can offer stronger light protection than a lighter green. The key is that olive is more sensitive to:
-
sulfur speciation,
-
redox pockets under the batch blanket,
-
and cullet organics that push reduction too far.
When reduction goes too far, olive can slide into brown, haze, or reboil issues. So olive needs “stable reduction,” not “more reduction.”
How the batch strategy differs (what gets changed on purpose)
Emerald:
-
Increase Cr₂O₃
-
Keep sulfur mainly as sulfate fining support
-
Keep redox stable to avoid yellowish shifts
Olive:
-
Use higher iron and a designed sulfur/redox plan
-
Allow more reduced sulfur behavior, but inside a controlled band
-
Expect stronger sensitivity to cullet contamination
| Feature | Emerald green | Olive green |
|---|---|---|
| Primary color driver | Cr₂O₃ + iron baseline | iron + reduced sulfur + some Cr |
| Redox style | stable, often less reducing | controlled mild reduction |
| Shade failure mode | yellowish-green drift or cords | brown drift, haze, reboil |
| Best cullet strategy | tight chromium and iron baseline | strong control of organics and sulfur inputs |
If a buyer needs emerald consistency, the plant must treat chromium dosing and mixing like a micro-ingredient program. If a buyer needs olive stability, the plant must treat redox and sulfur like a process state, not like a lab number.
What cullet percentage is optimal for cost control and stable green color in continuous furnaces?
Cullet cuts cost and energy. Still, cullet also adds variability. Green can handle more cullet than flint, but it still needs rules.
For many continuous green furnaces, a practical “sweet spot” is often around 40–65% cullet (and higher when cullet quality is strong). This range usually gives meaningful energy savings while keeping iron and redox variability manageable.

Why green can run higher cullet than flint
Green hides small iron swings. Flint does not. That means green can often accept higher recycled content without constant off-shade returns. Still, “accept” does not mean “ignore.” High cullet raises:
-
total iron,
-
contamination risk (ceramics, organics),
-
and redox noise.
So the optimal cullet rate is not a single number. It depends on how stable the cullet stream is.
Cost logic: cullet saves energy and raw materials
Cullet 8 melts easier than virgin batch. That reduces furnace energy demand and usually improves melt rate. The economic logic is simple: higher cullet can cut energy use and reduce raw material needs. The limit is usually not chemistry. The limit is quality.
Stability logic: cullet quality and color-family blending
For stable green, the cullet rules matter more than the percent:
-
Keep organics low (labels, inks, plastics)
-
Control ceramics/stone/porcelain (CSP)
-
Blend cullet lots to smooth iron swings
-
Avoid sudden shifts in flint/amber contamination levels
A good furnace-ready green cullet spec often allows some flint and amber in the stream. That helps supply, but the plant must manage the drift with blending and tracking.
A practical target plan that reduces risk
-
Start at 35–45% cullet if cullet supply is new or variable
-
Move toward 50–65% when cullet sorting and blending are stable
-
Push above that only when CSP and organics are tightly controlled and when redox monitoring is in place
| Cullet level | What you gain | What you risk | Best control point |
|---|---|---|---|
| 25–40% | easier stability | less cost saving | basic lot tracking |
| 40–65% | strong cost + energy wins | iron/redox drift | blending + redox discipline |
| 65%+ | max sustainability story | cords, brown drift, defects | tight cullet QA + live monitoring |
Green glass is a great place to push recycled content, but only if the cullet system is treated like a raw material spec, not like “free glass.”
How does a green formulation impact UV blocking and product shelf life for beverages and oils?
Many brands pick green because it feels “protective.” That is partly true, but the protection level depends on shade depth and bottle thickness.
Green glass reduces light exposure compared with clear glass, so it can slow light-driven quality loss in beverages and oils. Still, green usually protects less than amber, so shelf-life claims should be backed by transmission data, not assumptions.
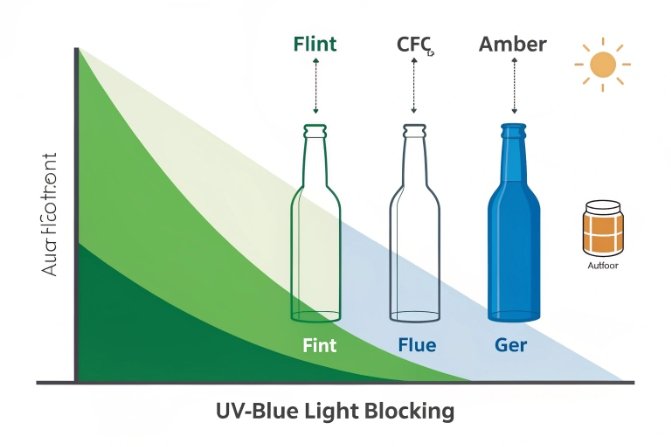
What green blocks, and what still gets through
Green bottles cut some UV and violet/blue light, depending on:
-
how deep the green is (iron + chromium level),
-
the iron valence (Fe²⁺ drives cooler deeper green),
-
and wall thickness.
For many beverages and oils, the key risk is light-driven oxidation and aroma changes. Less light means slower change. That is why wine and oil brands often avoid clear bottles.
Why olive green often protects better than lighter green
Olive green is usually darker and can involve a redox/sulfur balance that increases absorption in the harmful region. So olive often offers stronger protection than a lighter emerald-style green at the same thickness. Still, amber generally blocks more of the damaging band than most greens.
How I recommend buyers validate shelf-life benefit
A buyer should request a UV-Vis transmission 9 scan on real bottle wall thickness, not on a thin lab coupon. The scan should cover:
-
UVB (280–315 nm)
-
UVA (315–400 nm)
-
violet/blue (400–450 nm) for many beverage reactions
Then connect that scan to the product’s known sensitivity. For example, hop compounds in beer and many natural antioxidants in oils respond to high-energy light. So blocking short wavelengths matters more than blocking red light.
Keep the message honest: “improved,” not “fully protected”
Green can be a strong “middle choice” when the brand wants a premium look and better protection than clear. It is not always enough for the most sensitive formulas unless the green is quite dark or the bottle uses added UV-blocking design.
| Packaging color | Typical protection story | Best for | What to verify |
|---|---|---|---|
| Clear (flint) | minimal | products that are not light sensitive | baseline transmission |
| Green (emerald/Georgia) | moderate | beverages and oils needing some protection | UV-Vis 280–450 nm |
| Olive green | higher | beer, oils, light-sensitive beverages | UV-Vis + shelf simulation |
| Amber | strongest | very light-sensitive SKUs | UV-Vis + long-term stability |
Green formulation supports shelf life 10 when it is treated as a measurable performance feature. That means stable shade, documented transmission, and a cullet plan that does not drift.
Conclusion
Standard green bottles use a stable soda-lime base and tune green with iron valence and small Cr₂O₃ additions. Emerald and olive differ mostly by redox and sulfur strategy. Higher cullet works best with strong cullet QA and measured light transmission.
Footnotes
-
Soda-lime silica is the most common glass type for containers due to its chemical stability and workability. ↩
-
Redox control manages the oxidation state of the melt, directly influencing the final color of iron and chromium. ↩
-
Viscosity determines the working range of the glass melt, critical for forming bottles without defects. ↩
-
Annealing removes internal stresses in the bottle to prevent breakage after forming. ↩
-
Alumina is added to improve the chemical durability and resistance of the glass matrix. ↩
-
Light absorption properties of iron and chromium determine the specific shade and UV protection of the glass. ↩
-
Chromium oxide is the primary colorant for achieving deep, rich green hues in glass bottles. ↩
-
Using cullet reduces energy consumption and raw material costs in the glass manufacturing process. ↩
-
UV-Vis spectroscopy measures light transmission to verify the protective qualities of the colored glass. ↩
-
Proper glass formulation extends shelf life by protecting light-sensitive contents from photo-oxidation. ↩








