The problem is simple but costly. Light damages products. Many brands lose shelf life because they pick the wrong glass color.
Glass color directly controls which light wavelengths reach the product. Flint, amber, emerald, and cobalt glass each pass and block different parts of the spectrum, which affects shelf life, visual appeal, and label readability.

Light control is not only a design topic. It is a technical decision that affects stability, compliance, and consumer trust. Understanding spectra helps procurement teams avoid hidden risks and compare suppliers with real data by requesting a UV–Vis spectral transmission test report 1.
What spectra does flint (clear) glass pass or block?
Clear glass looks neutral, but it is not invisible to light.
Flint glass transmits most visible light and a portion of near-UV, which makes it the weakest option for light protection but the strongest for visibility and label clarity.
Why flint glass behaves this way
Flint glass contains very low levels of iron and other colorants. This reliance on low-iron raw materials 2 keeps absorption minimal. As a result, light passes through with little loss.
Key spectral behavior
- High transmittance from about 420–700 nm
- Partial transmission below 380 nm unless UV inhibitors are added
- Minimal absorption bands
Practical impact on products
Flint glass is often chosen for:
- Premium appearance
- Color-sensitive branding
- Products where clarity signals purity
But this choice carries risk.
Light below 450 nm triggers oxidation, vitamin loss, aroma breakdown, and color fade in many beverages and cosmetics. Clear glass offers almost no defense against this process.
Trade-off summary
| Aspect | Performance |
|---|---|
| UV protection | Very low |
| Visible clarity | Excellent |
| Label readability | Excellent |
| Shelf life support | Weak |
Flint glass works best when combined with secondary protection like cartons, UV coatings, or fast inventory turnover.
How does amber glass block UV and blue light?
Amber glass is the benchmark for light protection.
Amber glass blocks most UV and short-wavelength visible light below ~450 nm, which makes it the most effective color for preventing photodegradation.
Why amber glass is so effective
Amber glass uses a combination of iron, sulfur, and carbon 3. These form strong absorption bands in the UV and blue regions.
This absorption is not cosmetic. It is chemical.
Core absorption zones
- Near-zero transmission below 380 nm
- Strong reduction from 380–450 nm
- Gradual rise above 500 nm
Impact on shelf life
Many sensitive compounds degrade fastest under UV and blue light:
- Hops and iso-alpha acids
- Essential oils
- Vitamins A, B2, and C
- Natural pigments
Amber glass sharply slows these reactions.
Design and branding limits
The downside is visibility.
| Aspect | Performance |
|---|---|
| UV protection | Excellent |
| Visible clarity | Low |
| Label contrast | Medium |
| Premium appearance | Product-dependent |
Brands must rely more on label design and less on product color when using amber glass.
What protection do emerald (green) bottles really offer?
Green glass sits between flint and amber.
Emerald glass reduces UV and blue light but allows more short-visible wavelengths than amber, giving partial protection with better product visibility.
The chemistry behind green glass
Green glass usually contains iron and chromium. In many bottle programs, chromium oxide is used to intensify green shades 4, which shifts which wavelengths are absorbed compared with flint glass.
Typical spectral pattern
- Reduced UV below 380 nm
- Moderate reduction from 380–450 nm
- Strong transmission in green wavelengths
What this means in real use
Green glass works well when:
- The product needs some light protection
- Visual access still matters
- Branding favors natural or heritage cues
It is common in:
- Wine
- Olive oil
- Specialty beverages
Limitations to understand
Green glass does not block blue light completely. Products that are highly photosensitive may still degrade over time under retail lighting.
| Aspect | Performance |
|---|---|
| UV protection | Moderate |
| Visible clarity | Medium |
| Shelf life extension | Partial |
Emerald glass is a compromise, not a shield.
How does cobalt blue glass differ spectrally?
Cobalt glass is visually striking but technically misunderstood.
Cobalt blue glass blocks yellow and red wavelengths while allowing blue light to pass, which offers poor protection against photodegradation despite its dark appearance.
Why blue glass is risky
Cobalt oxide creates strong absorption in the red and yellow ranges. A practical way to understand this is that cobalt glass is used as an optical filter to mask yellow sodium emission 5—meaning it does not remove the damaging blue/violet region in the same way amber does.
Blue light is one of the most damaging ranges for many products.
Common misconceptions
Darker does not always mean safer.
Cobalt glass looks dense, but spectrally it:
- Allows 400–470 nm light
- Blocks less UV than amber
- Can accelerate degradation in some formulas
Where cobalt still works
Cobalt glass makes sense when:
- Marketing impact matters most
- Products are not light-sensitive
- Secondary packaging blocks light
It is often used in:
- Fragrances
- Decorative spirits
- Limited-edition SKUs
How do color choices affect shelf life and label readability?
Color choice forces a balance between protection and communication.
Darker glass improves shelf life but reduces product visibility, while lighter glass enhances branding but exposes contents to light damage.
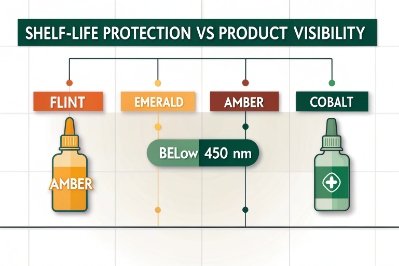
Shelf life impact
The largest transmittance drop happens below 450 nm. This is where:
- Amber excels
- Green helps
- Blue fails
- Flint exposes
This aligns with how the blue part of the visible spectrum (about 350–500 nm) 6 is especially efficient at driving light-struck reactions in sensitive beverages under exposure.
Thicker walls help, but color is more efficient than thickness for light control.
Label readability factors
Glass color affects:
- Contrast
- Color accuracy
- Metallic inks
- Backlighting effects
Clear and light green glass support high-contrast labels. Amber often requires bolder inks and stronger adhesives.
Combined effects
When color and thickness stack, protection increases fast. Thick amber shoulders can drop transmittance to near zero in critical bands.
How should procurement compare color batches using spectral data?
Visual inspection is not enough.
Procurement teams should compare glass color batches using spectral transmittance curves, not just color codes or samples.

What data to request
A proper data sheet should include:
- Wavelength range (280–800 nm)
- Percentage transmittance
- Sample thickness
- Measurement standard (for regulated or light-sensitive programs, many teams reference USP <671> Containers—Performance Testing 7 as a benchmarking framework)
What to compare
Focus on:
- UV cutoff point
- Average transmittance below 450 nm
- Batch-to-batch deviation
Why this matters
Color nonuniformity causes local transmittance variation. This means one bottle protects better than another from the same order.
Procurement should treat spectral limits like dimensional tolerances.
Are low-iron flint and tuned pigments narrowing the gap?
Technology is changing the rules.
Low-iron flint glass and tuned pigment systems are narrowing performance gaps, but they do not erase fundamental spectral differences.

Low-iron flint glass
Low-iron flint increases clarity. It does not improve protection. In fact, it often transmits more UV.
Tuned pigments
Modern color systems can:
- Sharpen absorption bands
- Improve color consistency
- Reduce unwanted transmission
But physics still applies.
Amber remains unmatched for UV and blue blocking. Green remains a compromise. Blue remains risky.
Smart combinations
The best results often come from:
- Moderate color
- Controlled thickness
- Secondary packaging
- Clear specifications
Conclusion
Glass color controls light, and light controls product life. Spectral data turns color from a design choice into a measurable performance decision.
Footnotes
-
Shows what a UV–Vis spectral transmission report includes and how results are evaluated across wavelengths. ↩︎ ↩
-
Explains how low-iron inputs produce clear bottle glass and why “clear” typically transmits more light. ↩︎ ↩
-
Summarizes how amber glass colorants create strong absorption below ~450 nm for high light protection. ↩︎ ↩
-
Provides practical industry context on iron/chromium contributions to green bottle color in wine and beverage packaging. ↩︎ ↩
-
Demonstrates cobalt glass filtering yellow light—useful for understanding why cobalt still passes damaging blue/violet wavelengths. ↩︎ ↩
-
Explains why blue/violet wavelengths are especially effective at causing light-struck reactions in sensitive beverages. ↩︎ ↩
-
A common benchmark chapter for light transmission expectations and reporting structure in regulated packaging evaluations. ↩︎ ↩








