Many buyers assume “glass = safe,” then get nervous when they hear about heavy metals, migration, or bottle breakage on the filling line.
Glass bottles protect liquid safety through chemical inertness, tight sealing systems, and strict compliance and testing that control migration, pressure, and sterility throughout the product’s life.

When we look at glass as a full system—material, closure, testing, and logistics—its value becomes clear: stable flavor, protected actives, and reliable shelf life for food, cosmetics, and pharmaceuticals.
Is glass chemically inert and suitable for sensitive formulations?
Formulators of serums, oils, syrups, and injections worry about two things: “Will the bottle contaminate my product?” and “Will my product attack the bottle?”
Glass is effectively inert for most foods, cosmetics, and pharma liquids. Its dense, non-porous surface creates a real barrier to gases and liquids and shows very low migration, especially with food-contact and pharma-grade compositions.
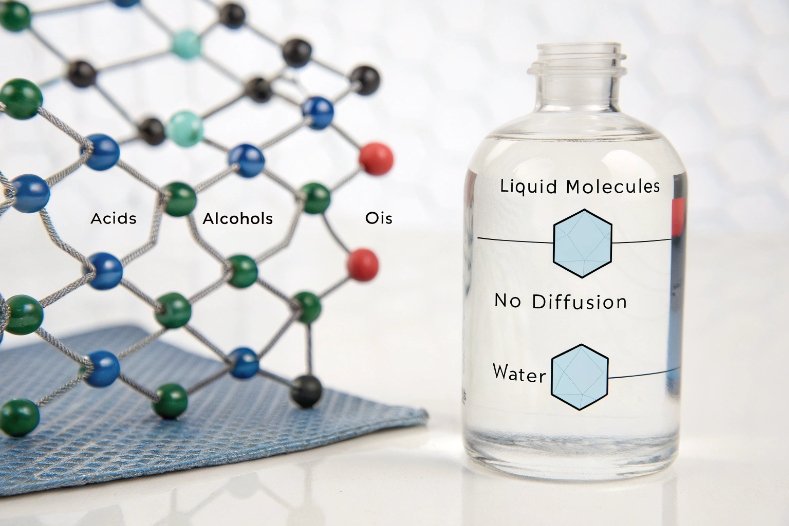
Why glass is so stable for liquids
Glass is an amorphous network mainly based on silica with other oxides 1. In soda-lime and borosilicate glass, the network is tight and non-porous. Molecules from the product cannot diffuse in, and components of the glass cannot easily diffuse out. This gives two big advantages:
-
Chemical inertness:
- Resistant to water, acids, alcohols, many oils, and most organic solvents.
- Does not carry plasticizers or volatile monomers.
- Properly formulated glass has extremely low levels of heavy metals and controlled leaching.
-
True barrier:
- Glass is practically impermeable to oxygen, CO₂, and vapor.
- No flavor scalping, no aroma loss, no solvent loss, and no external odor uptake.
Borosilicate glass (for example Type I pharma glass) 2 goes further. It has higher silica and boron content, lower alkali content, and shows even better resistance to leaching and pH changes. That is why it is used for injectables and sensitive biologics.
At the same time, glass tolerates high temperatures. It can go through hot-fill, pasteurization, retort processes, and, in pharma, steam sterilization and dry heat depyrogenation. These processes kill microbes and degrade pyrogens without damaging the container or releasing harmful substances, as long as the glass type is correctly chosen.
| Product Type | Typical Glass Choice | Key Safety Benefit |
|---|---|---|
| Carbonated drinks | Soda-lime beverage bottles | Strong, inert, gas-tight |
| Oils and essences | High-white or amber soda-lime | Aroma protection, light screening |
| Cosmetics serums | High-white, amber, or frosted | Low migration, premium appearance |
| Injectables | Type I borosilicate | Highest chemical durability, sterilizable |
| Syrups and elixirs | Type III / soda-lime | Good resistance, controlled leaching |
For most liquid products, glass offers the safest combination of low migration, strong barrier, and high processing temperature tolerance.
Which certifications and compliance tests (FDA, REACH, ISO) should suppliers provide?
Material safety is not just about what the supplier promises. Brand owners and regulators want documented proof that bottles and decorations are safe for food, cosmetics, and drugs.
For safe liquid packaging, suppliers should back their glass and closures with relevant FDA and EU food-contact statements, REACH and RoHS declarations, and quality system certifications such as ISO 9001, plus industry-specific standards like ISO 15378 for pharma.

Core regulatory and compliance documents
For food and beverage:
- FDA compliance (US) 3:
Suppliers should state that compositions and colorants comply with applicable FDA regulations for food-contact materials (for example, glass as an indirect food additive). Decorations like inks or coatings must also be food-contact safe where relevant. - EU food-contact compliance:
Declarations referencing EU Framework Regulation (EC) No 1935/2004 4 and relevant national or sector guidance (for example, for ceramics and glass) show that the material is suitable for contact with food. - REACH & RoHS declarations 5:
REACH (EU chemicals regulation) and RoHS (restriction of hazardous substances) declarations confirm that glass, coatings, and closures do not contain restricted substances above limits and that SVHC obligations are addressed.
For cosmetics and pharma:
- GMP and quality standards:
- ISO 9001: Quality management system for consistent production and traceability.
- ISO 15378: Specific for primary packaging materials for medicinal products (GMP requirements).
- Pharma glass types:
- USP / Ph. Eur. classifications (Type I, II, III) 6 define hydrolytic resistance and suitability for different injectable and non-injectable uses.
- Suppliers should provide test reports (hydrolytic resistance, internal surface tests) for vials and bottles intended for medicines.
Decorations and colors:
- Lead-free, cadmium-free, and other heavy-metal limits must be respected. Migration tests confirm that enamel inks, decals, and coatings stay within legal limits for food-contact surfaces.
Typical documentation pack a buyer should request
| Document / Certificate | Why It Matters |
|---|---|
| Declaration of compliance for food-contact | Confirms legal use for food/beverage |
| REACH & RoHS declaration | Confirms absence of restricted chemicals |
| Material data for glass & colors | Details of composition and limits |
| USP / Ph. Eur. test reports (for pharma) | Confirms hydrolytic and chemical resistance |
| ISO 9001 / ISO 15378 certificate | Shows controlled, traceable production |
| Migration / heavy metal test reports | Verifies safe levels under realistic conditions |
When these documents are ready on day one, it saves weeks of back-and-forth in approval projects and gives buyers confidence that the packaging will pass audits.
What closure systems and liners ensure leakproof, tamper-evident sealing?
Even the best glass loses its value if the closure leaks, lets in air, or can be opened and re-closed without visible trace. Seal quality is where liquid safety meets user trust.
Leakproof, tamper-evident performance comes from the combination of a well-formed glass finish, a suitable closure system (screw, crown, pump, dropper, etc.), and a compatible liner or gasket that matches the product’s chemistry and pressure.

Finish and closure types
Common systems include:
- Screw caps (continuous thread and lug caps):
Used widely on food jars, sauces, and cosmetics. The glass thread or lug profile must match the cap standard to ensure consistent torque and seal. - Crown caps and pry-off / twist-off finishes:
Classic for beer and carbonated beverages. These create an excellent gas-tight, tamper-evident seal when correctly applied. - Droppers, pumps, and mist sprayers:
Standard in cosmetics and pharma. They combine mechanical function with a sealing system, often with multiple gaskets. - Roll-on pilfer-proof (ROPP) caps:
Aluminum caps crimped onto glass threads, common on spirits and oils. The “pilfer band” gives clear tamper evidence.
In all cases, the glass finish must be dimensionally correct and properly annealed to avoid chipping and microcracks. A strong bottle with a damaged finish is still a weak package.
Role of liners and gaskets
The liner is the real interface between product and closure. Different liquids need different formulations:
- Foamed PE or EPE liners:
Good general-purpose option for oils, sauces, and dry products, with basic sealing and cushioning. - PVC / PVDC or co-extruded barrier liners:
Extra oxygen and aroma barrier for sensitive foods (for example, juices, coffee syrups) or pharmaceuticals. - Rubber or elastomer gaskets (NBR, EPDM, silicone):
Used in swing-top bottles, droppers, and pumps, chosen based on resistance to oils, alcohols, or aggressive actives. - Induction-seal liners (foil seals):
Provide tamper evidence and hermetic sealing, often used under screw caps on syrups and nutraceuticals.
Choosing the wrong liner can lead to swelling, hardening, or loss of elasticity. This causes slow leaks, flavor changes, or oxygen ingress.
Tamper-evident features
Tamper evidence protects both consumers and brand reputation. Options include:
- Breakable bridges on plastic screw caps (ring stays behind on first opening).
- Perforated or shrink sleeves over cap and neck.
- Vacuum “button” lids on hot-filled and pasteurized jars that stay depressed until opened.
- Tear-off bands on ROPP caps and metal closures.
| Feature | Safety Role |
|---|---|
| Correct finish dimensions | Allows consistent sealing torque |
| Compatible liner material | Prevents leaks and chemical interaction |
| Tamper band / pilfer ring | Signals first opening clearly |
| Foil / induction seal | Extra microbiological and leak protection |
| Shrink band or sleeve | Visual tamper evidence for retail |
Strong glass plus smart sealing ensures that liquids arrive in the same safe condition as when they left the filling line.
How are migration, pressure, and sterility tests used for quality assurance?
Safety is not a guess. It is measured and proven with tests that simulate storage, handling, and use. For liquid products, three key groups of tests stand out: migration, pressure/vacuum, and sterility or microbial tests.
Migration, pressure, and sterility testing confirm that glass and closures do not release harmful substances, that seals withstand carbonation or vacuum, and that microbial risks are controlled during processing and storage.

Migration and extractables
Migration tests check what, if anything, moves from packaging into the liquid under defined conditions (time, temperature, and simulant). For glass systems, this can cover:
- Global migration:
Total non-volatile material released into a food simulant (like ethanol, acetic acid, oil simulants). - Specific migration:
Focused tests for heavy metals (lead, cadmium, arsenic), elements like aluminum, or specific additives from coatings, inks, or closures. - Extractables and leachables (pharma) 7:
Stress tests at high temperature or long contact time with solvents or real drug products, followed by chemical analysis.
Results must stay below legal limits. Repeated testing over time makes sure no process changes introduce new risks.
Pressure, vacuum, and mechanical performance
For carbonated and hot-filled products, mechanical performance is as critical as chemistry.
Typical tests include:
- Internal pressure tests:
Bottles are pressurized with water or air to a set value above their working pressure (for example, for beer or soft drinks). The goal is to confirm a safety margin and identify weak designs or quality drifts. - Vacuum tests:
Hot-filled or pasteurized jars create internal vacuum when cooled. Vacuum-resistance tests check whether lids stay sealed and glass resists paneling or deformation. - Torque and seal integrity tests:
Measure the opening torque and check for leakage under pressure or during inverted storage.
These tests show whether the closure system, liner, and glass finish work together to protect the liquid against leaks and environmental ingress.
Sterility and microbiological safety
For food and beverages:
- Process validation of pasteurization, hot-fill, or retort regimes shows that the chosen time-temperature combination destroys target microorganisms.
- Regular microbiological testing of filled products (pathogens, spoilage organisms) confirms that cleaning, filling, and sealing are under control.
For pharmaceutical products:
- Sterility tests validate cleanroom filling, filtration, and container closure integrity.
- Container closure integrity tests (CCIT) 8—for example vacuum decay, helium leak, or dye ingress—show that the seal stays tight over shelf life.
Glass is a strong partner here because:
- It withstands high sterilization temperatures.
- It does not deform during heat cycles, so seal geometry stays stable.
- It does not release plasticizers or other volatile compounds during sterilization.
| Test Type | What It Confirms |
|---|---|
| Migration / leachables | Chemical safety and low contamination |
| Pressure / vacuum | Resistance to carbonation or hot-fill processes |
| Torque / seal | Leakproof, consistent sealing |
| Sterility / CCIT | Microbiological safety over shelf life |
When these tests are built into a supplier’s quality system, every batch of bottles becomes a controlled packaging component, not just “empty glass.”
Conclusion
Glass bottles keep liquids safe by combining inert material, smart closures, and proven testing so products stay pure, sealed, and stable from filling line to final use.
Footnotes
-
Background on how glass’s amorphous silica network drives its chemical and mechanical stability. ↩︎ ↩
-
Overview of pharmaceutical Type I borosilicate glass and its superior hydrolytic resistance. ↩︎ ↩
-
FDA resource on food-contact substances and packaging safety for US markets. ↩︎ ↩
-
Full legal text of EU Framework Regulation 1935/2004 on food-contact materials. ↩︎ ↩
-
Explanation of REACH duties and restricted substances affecting packaging materials. ↩︎ ↩
-
USP chapter on glass containers, including Type I, II, and III classification tests. ↩︎ ↩
-
FDA guidance on assessing extractables and leachables risks in pharmaceutical products. ↩︎ ↩
-
FDA guidance on container closure integrity testing for sterile injectable drug products. ↩︎ ↩








